Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Outlook 2025: Mapping Growth, Innovation, and Regional Shifts
Claim your 30% discount on Global Market Reports with code ONLINE30. Limited time only.
What Are the Key Milestones in the Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market’s Growth Trajectory From 2025 To 2034?
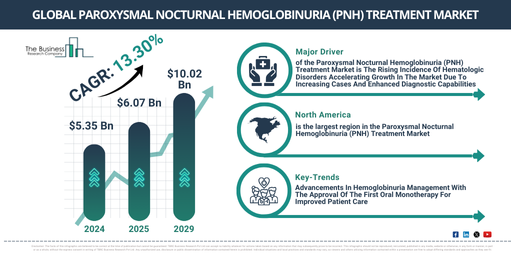
In recent years, the paroxysmal nocturnal hemoglobinuria (pnh) treatment market has seen substantial growth. The market size is predicted to increase from $5.35 billion in 2024 to $6.07 billion in 2025, with a compound annual growth rate (CAGR) of 13.5%. This notable growth during the historic period can be ascribed to the rising occurrences of neurological disorders and chronic pain conditions, enhanced awareness of the disease, increasing instances of paroxysmal nocturnal hemoglobinuria (PNH), heightened knowledge about rare blood disorders, and the rise in regulatory approvals along with research and development (R&D).
Over the coming years, the market size for paroxysmal nocturnal hemoglobinuria (pnh) treatment is anticipated to experience substantial growth, swelling to$10.02 billion by 2029 with a Compound Annual Growth Rate (CAGR) of 13.3%. This predicted growth within the forecast period can be ascribed to the rising incidence of uncommon ailments, heightened health care expenditure and access to cutting-edge treatments, incremented investment in health care facilities, an upswing in home-based examinations and remote monitoring, along with expanded research and development undertakings. The forecast period is also expected to observe major trends such as advancements in genetic testing and biomarkers, improvements in therapies, creation of targeted therapies, progression in biotechnology, and the development of innovative treatments.
Download a free sample to assess the report’s scope and structure:
https://www.thebusinessresearchcompany.com/sample.aspx?id=23469&type=smp
What External and Internal Drivers Are Contributing to the Growth of theParoxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
The rise in hematologic disorders is anticipated to stimulate the paroxysmal nocturnal hemoglobinuria (PNH) treatment market’s expansion. Hematologic disorders encompass a wide array of health issues that harm the blood and blood-forming organs. These include irregularities in red and white blood cells, platelets, bone marrow, and the coagulation system, potentially leading to anemia, bleeding problems, or cancers. Factors such as aging demographics and genetic susceptibilities primarily fuel the surge in hematologic disorders, with genetic anomalies playing a pivotal role in triggering abnormal blood cell formation, which induces numerous hematologic diseases. The treatment of paroxysmal nocturnal hemoglobinuria (PNH) effectively manages hematologic disorders by providing precise therapies that lessen complement-mediated hemolysis, curtail thrombotic issues, and enhance patient survival chances. As per Cancer Research UK, a charity organization based in the UK, the average number of new leukemia cases (a hematologic disorder) in the UK is projected to rise from roughly 12,400 in 2023-2025 to an estimated 14,500 by 2038-2040. Hence, the escalating incidence of hematologic disorders stimulates the growth of the paroxysmal nocturnal hemoglobinuria (PNH) treatment market.
What Segment Types Define the Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Structure?
The paroxysmal nocturnal hemoglobinuria (pnh) treatmentmarket covered in this report is segmented –
1) By Treatment Type: Complement C5 Inhibitors; Anti-Complement Component 5a Receptor (C5aR) Monoclonal Antibodies; Alternative Pathway Inhibitors; Hematopoietic Stem Cell Transplantation; Other Therapies
2) By Route Of Administration: Intravenous; Subcutaneous; Oral
3) By Market Availability: Approved; In Pipeline; Investigational
4) By End-Use: Hospitals; Specialty Clinics; Homecare Settings; Other End-Users
Subsegments:
1) By Complement : C5 InhibitorsEculizumab (Soliris); Ravulizumab (Ultomiris); Emerging C5 Inhibitors; Biosimilars Of C5 Inhibitors,
2) By Anti-Complement Component 5a Receptor Monoclonal Antibodies: Avacopan (Tavneos); Other Investigational C5aR mAbs; Monotherapy vs Combination Therapy; Clinical Trial Phase (Phase I, II, III)
3) By Alternative Pathway Inhibitors: C3 Inhibitors; Factor B Inhibitors; Factor D Inhibitors; Targeting Complement Amplification; Combination Therapies And Dual Inhibition
4) By Hematopoietic Stem Cell Transplantation (HSCT) : Autologous HSCT vs. Allogeneic HSCT; Pre-Transplant Conditioning Therapies; Post-Transplant Immunosuppressive Therapies
5) By Other Therapies: Immunosuppressive Therapy; Supportive Care; Anticoagulation Therapy; Gene Therapy
Request customized data on this market:
https://www.thebusinessresearchcompany.com/customise?id=23469&type=smp
Which Geographic Areas Hold the Strongest Growth Potential in the Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
North America was the largest region in the paroxysmal nocturnal hemoglobinuria (PNH) treatment market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the paroxysmal nocturnal hemoglobinuria (PNH) treatment market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
What Long-Term Trends Are Transforming the Competitive Landscape of the Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
Leading businesses within the paroxysmal nocturnal hemoglobinuria (PNH) treatment market are concentrating their efforts on crafting groundbreaking solutions, such as oral factor B inhibitors. These innovative treatments aim to enhance the effectiveness of therapies for PNH and simplify patient handling of the disorder. Oral Factor B inhibitors are precision drugs, geared towards selectively blocking Factor B in the alternative complement pathway, with the goal of managing the overactive immune system and decreasing inflammation linked to complement-mediated conditions. For example, in December 2023, Novartis AG, a pharmaceutical corporation hailing from Switzerland, unveiled its FDA-approved Fabhalta (iptacopan), the first-ever oral monotherapy for PNH treatment. This ground-breaking Factor B inhibitor offers a holistic approach to managing complement-mediated hemolysis by pinpointing the alternative complement pathway in its early stages, thereby enhancing hemoglobin concentrations and lessening the need for blood transfusions. The green light for Fabhalta signifies a revolutionary leap forward in PNH treatment by offering superior effectiveness, improved patient adherence, and a substitute for intravenous therapies, revolutionizing the treatment scene for this unusual hematologic condition.
View the full report here:
What Is the Definition of the Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
Paroxysmal nocturnal hemoglobinuria (PNH) treatment refers to medical therapies to manage PNH, a rare blood disorder characterized by hemolysis (destruction of red blood cells), blood clots, and bone marrow dysfunction. Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, acquired hematologic disorder characterized by the destruction of red blood cells due to complement-mediated hemolysis, bone marrow dysfunction leading to cytopenias, and an increased risk of thrombosis. Paroxysmal nocturnal hemoglobinuria (PNH) management focuses on inhibiting complement activation to prevent hemolysis, addressing bone marrow failure, and mitigating thrombotic complications to improve patient outcomes and quality of life.
Purchase the full report and get a swift delivery:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=23469
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model