Global Polyether Ether Ketone (PEEK) Interbody Devices Market Growth Prospects and Strategic Opportunities Through 2029
Claim your 20% discount on Global Market Reports with code ONLINE20. Limited time only.
What Are The Market Size Estimates For The Polyether Ether Ketone (PEEK) Interbody Devices Market?
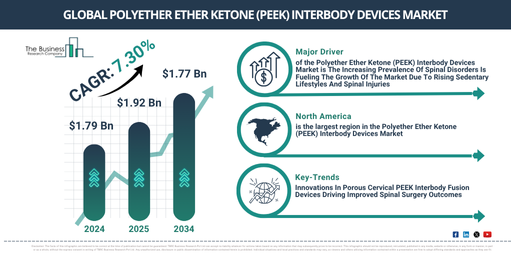
There has been a significant growth in the market size of polyether ether ketone (PEEK) interbody devices in recent years. It is anticipated to expand from a worth of $1.79 billion in 2024 to $1.92 billion in 2025, exhibiting a compound annual growth rate (CAGR) of 7.6%. The factors contributing to its growth during the historic period include a rising utilization in non-medical industries, an uptick in medical tourism, a surge in demand for MRI-compatible implants, improved clinical outcomes, and a growing prevalence of chronic back pain cases.
In the next few years, a significant increase is anticipated in the market size of polyether ether ketone (PEEK) interbody devices. The market value is projected to reach $2.56 billion by 2029, with a compound annual growth rate (CAGR) of 7.3%. The escalation during the forecast period could be linked to an increase in spinal disorders, a rise in vehicle production, the expansion of the automotive industry, a surge in healthcare spending, and the heightened use of hybrid material devices. The primary trends observed during this period are likely to be enhancements in technology, progress in medical technology, the uptake of advanced spinal implants, the inventive application of PEEK technology, and the introduction of sophisticated surgical techniques.
Get A Free Sample Of The Report:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25879&type=smp
Which Growth Drivers Are Shaping The Polyether Ether Ketone (PEEK) Interbody Devices Market Outlook?
The growing occurrence of spinal disorders is anticipated to stimulate the expansion of the PEEK interbody devices market. Spinal disorders are medical issues impacting the spine, which encompasses bones, discs, nerves, and muscles. These disorders are surging due to stationary lifestyles, where extended periods of sitting and diminished physical activity results in weakened spinal support muscles and intensified strain on the spine. PEEK interbody devices assist in the treatment of these disorders by providing robust, yet flexible support between vertebrae, enhancing spinal stability, encouraging natural bone growth, and retaining imaging clarity during subsequent scans. For example, Spinal Cord Inc., a US-centric resource for individuals having spinal cord injuries and their families, reported approximately 18,000 fresh cases of spinal cord injuries (SCI) in May 2023 in the USA. Around 302,000 individuals were expected to be living with SCI countrywide. As a result, the ascending prevalence of spinal ailments is propelling the expansion of the PEEK interbody devices market.
What Are The Primary Segmentation Parameters In The Polyether Ether Ketone (PEEK) Interbody Devices Market?
The polyether ether ketone (peek) interbody devices market covered in this report is segmented –
1) By Product Type: Posterolateral Fusion Devices, Interbody Fusion Devices, Anterior Lumbar Interbody Fusion Devices (Alif), Extreme Lateral Interbody Fusion Devices (Xlif), Posterior Lumbar Interbody Fusion Devices (Plif), Transforaminal Lumbar Interbody Fusion Devices (Tlif), Other Devices
2) By Application: Spinal Fusion Surgery, Degenerative Disc Disease, Spinal Trauma, Spinal Deformity
3) By Distribution Channel: Direct Sales, Online Sales, Third-Party Distributors
4) By End-User: Hospitals And Clinics, Ambulatory Surgery Clinics, Other End-Users
Subsegments:
1) By Posterolateral Fusion Devices: Single-Level Devices, Multi-Level Devices, Expandable Devices
2) By Interbody Fusion Devices: Cervical Fusion Devices, Lumbar Fusion Devices, Hybrid Devices
3) By Anterior Lumbar Interbody Fusion Devices (ALIF): Flat-Profile Devices, Curved-Profile Devices, Cage-Style Devices
4) By Extreme Lateral Interbody Fusion Devices (XLIF): Plated Devices, Cage-Style Devices, Oblique Devices
5) By Posterior Lumbar Interbody Fusion Devices (PLIF): Lordotic Devices, Flat-Cage Devices, Mesh Or Porous Devices
6) By Transforaminal Lumbar Interbody Fusion Devices (TLIF): Hollow Devices, Expandable Cages, Radiolucent Devices
7) By Other Devices: Miniature Interbody Fusion Devices, Biocomposite Devices, Modular Devices
Which Emerging Trends Are Reshaping The Polyether Ether Ketone (PEEK) Interbody Devices Market Landscape?
Leading enterprises in the PEEK interbody devices market are channeling their efforts into the creation of innovative products such as porous cervical IBF systems. These systems aim to augment osseointegration, bolster implant stability, and expedite patient recovery. The porous cervical IBF, or interbody fusion system, is a medical implant employed in spinal surgeries, particularly for cervical spine fusion operations. The purpose of this system is to substitute damaged or degenerated cervical intervertebral discs and to foster bone growth among neighboring vertebrae, consequently ensuring spinal stability. For example, in August 2022, Innovasis, a medical device company based in the US, gained 510(k) clearance from the US Food and Drug Administration for its product, HAcancellous PEEK-C, the pioneer hydroxyapatite (HA) porous PEEK interbody fusion device contrived for cervical spine procedures. This device is intended for single-level anterior cervical discectomy and fusion (ACDF) procedures from C2 to T1 in skeletally mature patients suffering from degenerative disc disease. It merges a porous structure with hydroxyapatite (HA) to facilitate bone growth and speed up fusion. The introduction of this device is expected to enhance spinal fusion outcomes by promoting superior bone integration and hastening fusion.
Which Organizations Are At The Forefront Of The Polyether Ether Ketone (PEEK) Interbody Devices Market?
Major companies operating in the polyether ether ketone (peek) interbody devices market are Medtronic Inc., Stryker Corporation, Zimmer Biomet Holdings Inc., Globus Medical Inc., NuVasive Inc., Orthofix Medical Inc., Exactech Inc., Xtant Medical Holdings Inc., Alphatec Spine Inc., Spinal Elements Inc., Providence Medical Technology Inc., FossiLabs LLC, Life Spine Inc., Curiteva Inc., Medacta International S.A., LDR Holding Corporation, Cavendish Implants Limited, SINTX Technologies Inc., In2Bones Global Inc., and Xilloc Medical International B.V.
Access The Complete Report Here:
Which Region Holds The Greatest Opportunity For Polyether Ether Ketone (PEEK) Interbody Devices Market Expansion?
North America was the largest region in the polyether ether ketone (PEEK) interbody devices market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the polyether ether ketone (peek) interbody devices market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Customize Your Report Here:
https://www.thebusinessresearchcompany.com/customise?id=25879&type=smp
Browse Through More Reports Similar to the Global Polyether Ether Ketone (PEEK) Interbody Devices Market 2025, By The Business Research Company
Interbody Fusion Cages Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/interbody-fusion-cages-global-market-report
Micro Injection Molded Plastic Global Market Report 2025
Exterior Automotive Plastics Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/exterior-automotive-plastics-global-market-report
About The Business Research Company:
With over 17500+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model