Adeno-Associated Virus (AVV) Vectors In Gene Therapy Industry Overview 2025: Growth Projections and Market Trends
Unlock Global Insights – Save 20% on Market Reports with Code ONLINE20
What Is The Future Market Size Estimation For The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Covering 2025–2034?
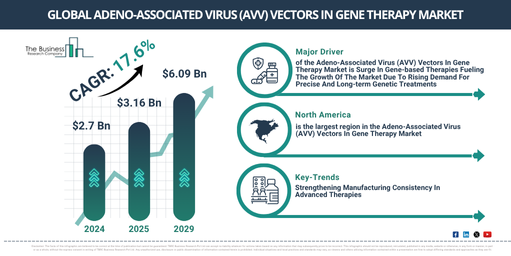
The adeno-associated virus (avv) vectors in gene therapy market has witnessed rapid growth in recent years. It is anticipated to increase from $2.7 billion in 2024 to $3.16 billion in 2025, exhibiting a compound annual growth rate (CAGR) of 16.7%. This historical expansion can be attributed to augmented investment in gene therapy research, the growing embrace of gene editing technologies, a surge in genetic disorders and unaddressed medical requirements, heightened demand for targeted therapies, and an uptick in collaborations between biotechnology companies and research institutions.
The adeno-associated virus (AVV) vectors in gene therapy market size is projected for substantial expansion in the coming years, reaching $6.09 billion by 2029 with a compound annual growth rate (CAGR) of 17.6%. This anticipated growth is driven by rising advancements in AAV capsid engineering, an increasing focus on precision medicine, growing investment in genetic research and biotechnology, a rise in gene therapy clinical trials and approvals, and increasing demand for personalized treatments. Major trends foreseen for this period include advancements in artificial intelligence applied to AAV capsid engineering, the development of hybrid AAV vectors designed for larger genetic payloads, innovation in bispecific antibody-mediated AAV targeting, progress in scalable AAV production and purification technologies, and the creation of flexible AAV platforms that support diverse viral vectors.
Unlock Your Free Sample Report for Exclusive Market Data:
https://www.thebusinessresearchcompany.com/sample.aspx?id=24135&type=smp
What Are The Emerging Market Drivers Creating New Opportunities In The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Industry?
A growing emphasis on gene-based therapies is anticipated to drive the expansion of adeno-associated virus (AAV) vectors within the gene therapy market in the future. Such therapies involve modifying or manipulating genes to address, treat, or prevent diseases at a molecular level. Progress in genetic research fuels this heightened focus on developing gene-based therapies, as these advancements boost the precision and efficacy of gene editing methods. Adeno-associated virus (AAV) vectors in gene therapy contribute to the creation of gene-based therapies by ensuring the safe and efficient delivery of therapeutic genes into target cells, thanks to their low immunogenicity and capacity for sustained gene expression. For example, data from IQVIA, a US-based provider of advanced analytics and technology solutions for the life sciences industry, indicated that global expenditure on cell and gene therapies reached $5.9 billion in 2023, marking a 38% rise compared to 2022. Consequently, the expanding focus on developing gene-based therapies is stimulating the growth of the adeno-associated virus (AAV) vectors market.
What Are The Major Segments Identified In The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market?
The adeno-associated virus (avv) vectors in gene therapy market covered in this report is segmented –
1) By Type of Therapy: Gene Augmentation, Immunotherapy, Other Type of Therapy
2) By Type of Gene Delivery Method Used: Ex Vivo, In Vivo
3) By Scale of Operation: Preclinical, Clinical, Commercial
4) By Target Therapeutic Area: Genetic Disorders, Hematological Disorders, Infectious Diseases, Metabolic Disorders, Ophthalmic Disorders, Muscle Disorders, Neurological Disorders, Other Target Therapeutic Area
Subsegments:
1) By Gene Augmentation: Monogenic Disorders, Neurological Disorders, Muscular Disorders, Ophthalmological Disorders, Metabolic Disorders
2) By Immunotherapy: Oncology, Infectious Diseases, Autoimmune Disorders, Vaccine Development, T-Cell Engineering
3) By Other Type of Therapy: Gene Editing Support, RNA Interference, Neuroprotection and Neuroregeneration, Anti-Inflammatory Applications, Regenerative Medicine
What Trends Are Redefining The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Dynamics?
Major companies engaged in the adeno-associated virus (AAV) vectors within the gene therapy market are concentrating on advanced innovations, notably tailored vector diversity for assay versatility. This approach aims to enhance target tissue specificity, improve therapeutic efficacy, and accelerate the development of customized gene therapies for a wide range of indications. Tailored vector diversity for assay versatility refers to the strategic use of multiple AAV serotypes or constructs to support diverse analytical tests and therapeutic applications. For instance, in May 2024, Charles River Laboratories, a US-based pharmaceutical company, introduced new reference materials for adeno-associated virus (AAV) and lentiviral vectors (LVV). These materials are intended to support the expanding requirements of cell and gene therapy (CGT) development. The comprehensive portfolio facilitates a smoother transition from early-stage research to GMP-grade production, helping to standardize processes and improve consistency in viral vector manufacturing. This launch effectively addresses a critical challenge in scaling CGT programs towards clinical and commercial readiness.
Which Leading Companies Dominate The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Share?
Major companies operating in the adeno-associated virus (AVV) vectors in gene therapy market are F. Hoffmann-La Roche Ltd., Sanofi S.A., Novartis AG, Astellas Gene Therapies Inc., Biogen Inc., Sarepta Therapeutics Inc., Oxford BioMedica plc, Rocket Pharmaceuticals Inc., Aldevron LLC, REGENXBIO Inc., uniQure N.V., Passage Bio Inc., Voyager Therapeutics Inc., Dyno Therapeutics Inc., MeiraGTx Holdings plc, Abeona Therapeutics Inc., 4D Molecular Therapeutics Inc., GenSight Biologics S.A., Taysha Gene Therapies Inc., LogicBio Therapeutics Inc.
Download The Full Report For Exclusive Market Findings:
What Regional Growth Trends Are Shaping The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Outlook?
North America was the largest region in the adeno-associated virus (AVV) vectors in gene therapy market in 2024. The regions covered in the adeno-associated virus (AVV) vectors in gene therapy market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Version Of The Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market Report:
https://www.thebusinessresearchcompany.com/customise?id=24135&type=smp
Browse Through More Reports Similar to the Global Adeno-Associated Virus (AVV) Vectors In Gene Therapy Market 2025, By The Business Research Company
Gene Editing Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/gene-editing-global-market-report
Nucleic Acid Based Gene Therapy Global Market Report 2025
Genes Therapy Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/genes-therapy-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model