Mobile Health (mHealth) Adverse-Event Reporting Apps Market Forecast: What to Expect by 2029
Unlock Global Insights – Save 20% on Market Reports with Code ONLINE20
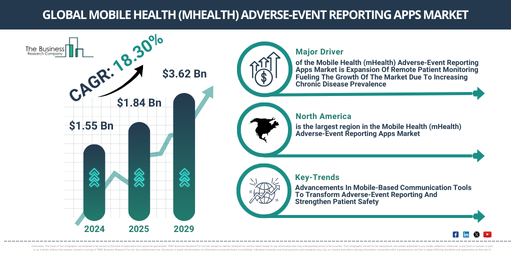
What Are The Forecasted Market Size Estimates For The Mobile Health (mHealth) Adverse-Event Reporting Apps Market Between 2025 And 2029?
The mobile health (mHealth) adverse-event reporting apps market has experienced rapid expansion recently. Projections indicate its size will increase from $1.55 billion in 2024 to $1.84 billion in 2025, demonstrating a compound annual growth rate (CAGR) of 18.7%. Historically, this growth was driven by factors such as regulatory initiatives promoting pharmacovigilance compliance, increasing adoption of smartphones and internet, greater patient involvement in healthcare, heightened awareness of adverse drug reactions, and the broadening of digital health ecosystems.
The mobile health (mHealth) adverse-event reporting apps market size is anticipated to experience substantial growth over the next few years. It is projected to expand to $3.62 billion by 2029, achieving a compound annual growth rate (CAGR) of 18.3%. This expansion during the forecast period is attributable to the integration with electronic health records (EHR/EMR), rising adoption of remote patient monitoring, supportive government digital health initiatives, an increased focus on real-time drug safety surveillance, and the growing demand for personalized and patient-centric healthcare. Significant trends expected in the forecast period include artificial intelligence-driven adverse event detection, blockchain-enabled data security, cloud-based reporting platforms, integration with wearable devices, and natural language processing for patient feedback.
Unlock Your Free Sample Report for Exclusive Market Data:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28273&type=smp
Which Drivers Are Impacting The Mobile Health (mHealth) Adverse-Event Reporting Apps Market?
The projected expansion of remote patient monitoring is anticipated to fuel the growth of the mobile health (mHealth) adverse-event reporting apps market in the future. Remote patient monitoring (RPM) involves using digital technologies to gather medical and health-related data from patients outside traditional healthcare environments. The increase in RPM is largely attributed to the rising prevalence of chronic diseases, which creates a demand for continuous, real-time patient monitoring beyond conventional healthcare settings to enhance outcomes and reduce hospital visits. Remote patient monitoring supports the advancement of mobile health (mHealth) adverse-event reporting apps by enabling ongoing collection of patient health data, facilitating prompt reporting and tracking of adverse drug reactions or treatment side effects through mobile platforms. For instance, in August 2023, according to Vivalink, a US-based healthcare technology company, 81% of clinicians now utilize remote patient monitoring (RPM), a significant jump from just 20% in 2021, representing a 305% increase. Furthermore, 84% of current RPM users intend to expand their use of the technology in 2024. Consequently, the expansion of remote patient monitoring is a key driver for the growth of the mobile health (mHealth) adverse-event reporting apps market.
How Is The Mobile Health (mHealth) Adverse-Event Reporting Apps Market Classified Into Different Segments?
The mobile health (mhealth) adverse-event reporting apps market covered in this report is segmented as
1) By App Type: Standalone Adverse-Event Reporting Apps, Integrated mHealth Platforms
2) By Platform: Android, iPhone Operating System (IOS), Web-Based
3) By Application: Pharmacovigilance, Clinical Trials, Patient Safety Monitoring, Other Applications
4) By End-User: Hospitals And Clinics, Pharmaceutical Companies, Research Organizations, Patients, Other End Users
Subsegments:
1) By Standalone Adverse-Event Reporting Apps: Patient-Centric Reporting Apps, Healthcare Provider Reporting Apps, Regulator-Focused Reporting Apps, Pharmacovigilance Reporting Apps
2) By Integrated mHealth Platforms: Electronic Health Record or Electronic Medical Record Integrated Reporting Modules, Telemedicine Platform-Integrated Reporting, Clinical Trial and Research Platform Integration, Hospital and Pharmacy Management System Integration
How Are Trends Influencing The Mobile Health (mHealth) Adverse-Event Reporting Apps Market?
Leading companies in the mobile health (mHealth) adverse-event reporting apps market are concentrating on developing advanced solutions, such as mobile-based communication tools, to speed up adverse-event detection and improve overall drug safety monitoring. Mobile-based communication tools are digital platforms or applications that facilitate seamless interaction, reporting, and information sharing through smartphones or tablets, allowing real-time connectivity between patients, healthcare providers, and regulators for quicker decision-making and better healthcare outcomes. For instance, in September 2023, Bharat Serums and Vaccines Limited (BSV), an India-based biopharmaceutical company, introduced the BSV AER, a new mobile-based communication application. This app permits healthcare professionals, consumers, and company personnel to electronically report potential adverse events from BSV products using smartphones or tablets anywhere in India, at no cost. BSV AER is directly integrated with BSV’s pharmacovigilance system, thereby streamlining the timely reporting and evaluation of adverse events to support patient safety through efficient management and swift responses.
Who Are The Core Companies Influencing The Mobile Health (mHealth) Adverse-Event Reporting Apps Market Landscape?
Major companies operating in the mobile health (mhealth) adverse-event reporting apps market are IQVIA Holdings Inc., Veeva Systems Inc., Itransition Group Inc., Bharat Serums and Vaccines Limited, ArisGlobal LLC, Saama Technologies Inc., Optimal Strategix Group Inc., OSP Labs Private Limited, K Health Inc., SafetyCall International PLLC, Medwatcher, Healthy.io Ltd., InPhase Technologies Ltd., Tiga Healthcare Technologies Inc., Qualityze Inc., iVigee Services AS, Advera Health Analytics Inc., Preventice Solutions Inc., Luscii Healthtech BV, and My Mhealth Limited.
Download The Full Report For Exclusive Market Findings:
What Are The Key Regional Factors Influencing The Mobile Health (mHealth) Adverse-Event Reporting Apps Market Growth?
North America was the largest region in the mobile health (mHealth) adverse event reporting apps market in 2024. The regions covered in mobile health (mHealth) adverse event reporting apps report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East and Africa.
Request A Customized Version Of The Mobile Health (mHealth) Adverse-Event Reporting Apps Market Report:
https://www.thebusinessresearchcompany.com/customise?id=28273&type=smp
Browse Through More Reports Similar to the Global Mobile Health (mHealth) Adverse-Event Reporting Apps Market 2025, By The Business Research Company
Mhealth Apps Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/mhealth-apps-global-market-report
Disease Management Apps Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/disease-management-apps-global-market-report
Womens Health App Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/womens-health-app-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model