How Will The Oncology Companion Diagnostics Market Expand At A CAGR Of 12% Through 2029?
Claim your 20% discount on Global Market Reports with code ONLINE20. Limited time only.
What Is The Present And Forecasted Size Of The Oncology Companion Diagnostics Market?
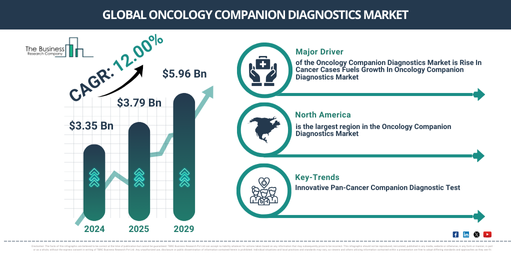
The market size for oncology companion diagnostics has witnessed a significant expansion in the recent past. The market, which stood at $3.35 billion in 2024, is expected to reach a value of $3.79 billion in 2025, growing at a compound annual growth rate (CAGR) of 13.2%. Factors contributing to this growth during the historic period include a rise in adverse drug reaction cases, heightened demand for inclusive genomic testing, increasing worldwide cancer cases, growing need for targeted therapies, and an increase in the number of clinical trials.
The size of the oncology companion diagnostics market is projected to expand considerably in the coming years, reaching $5.96 billion in 2029 with a Compound Annual Growth Rate (CAGR) of 12.0%. The anticipated growth during the forecast period is mainly due to the increasing relevance of personalized medicine, a rise in diagnostic tests, a heightened emphasis on customized cancer treatments, escalated healthcare spending, and growing incidence and awareness of cancer. Major developments estimated for the forecast period involve improvements in diagnostic technologies, next-generation sequencing (NGS), the incorporation of artificial intelligence (AI), advancements in liquid biopsy technologies, the emergence of point-of-care companion diagnostic devices, and cutting-edge testing technologies.
Get A Free Sample Of The Report:
https://www.thebusinessresearchcompany.com/sample.aspx?id=15623&type=smp
What Drivers Are Accelerating Expansion Of The Oncology Companion Diagnostics Market?
The escalation of cancer instances is projected to stimulate the expansion of the oncology companion diagnostics market in the future. Cancer denotes a heterogeneous group of conditions caused by anomalous cells in the body that proliferate and spread unchecked. The surge in cancer cases can be attributed to lifestyle changes, environmental influences, the obesity crisis, tobacco consumption, and genetic determinants. Oncology companion diagnostics play a pivotal role in precision medicine strategies in oncology, transforming cancer care by facilitating personalized treatment selection, boosting treatment effectiveness, reducing unwanted side effects, easing enrollment in clinical trials, and improving treatment tracking. For instance, as per May 2024 data from the National Cancer Institute, a government agency based in the US, approximately 20 million new cancer cases and 9.7 million cancer-associated deaths were registered annually in 2022, and forecasts suggest that by 2040, the numbers would escalate to 15.3 million fatalities and 29.9 million novel cases each year. Consequently, the escalation in cancer instances fuels the expansion of the oncology companion diagnostics market.
What Are The Primary Segmentation Parameters In The Oncology Companion Diagnostics Market?
The oncology companion diagnosticsmarket covered in this report is segmented –
1) By Product: Instrument; Consumables; Software
2) By Disease Type: Melanoma; Breast Cancer; Colorectal Cancer; Leukemia; Prostate Cancer; Other Disease Types
3) By Technology: Immunohistochemistry (IHC); In Situ Hybridization (ISH) Or Fluorescence In Situ Hybridization (FISH); Polymerase Chain Reaction (PCR); Next-Generation Sequencing (NGS); Other Technologies
4) By End User: Hospital; Pathology Or Diagnostic Laboratory; Academic Medical Center; Other End-Users
Subsegments:
1) By Instrument: Polymerase Chain Reaction (Pcr) Instruments; Next-Generation Sequencing (Ngs) Systems; Immunohistochemistry (Ihc) Instruments; Fluorescence In Situ Hybridization (Fish) Instruments; Microarray Platforms
2) By Consumables: Reagents And Kits (Dna Or Rna Extraction Kits, Pcr Reagents); Biopsy Needles; Testing Slides And Chips; Control And Calibration Samples
3) By Software: Data Analysis Software; Bioinformatics Software ( For Gene Expression Analysis); Companion Diagnostic Decision Support Software; Clinical Workflow Management Software
Which Emerging Trends Are Reshaping The Oncology Companion Diagnostics Market Landscape?
Leading firms in the oncology companion diagnostics market are honing in on the creation of groundbreaking products, including in vitro diagnostic tests, to maintain their market standing. In vitro diagnostic tests, also known as IVD tests, involve clinical tests performed on human body samples, such as tissue or blood samples, in a controlled environment typically in a lab. As an example, in May 2022, biotech company based in the US, Illumina Inc., introduced a new pan-cancer companion diagnostic (CDx) function to the in-vitro diagnostic TruSight Oncology (TSO) Comprehensive (EU) test. This diagnostic procedure provides a detailed tumor profile by analyzing various tumor genes and biomarkers, equipping healthcare practitioners with the necessary information to decide on targeted cancer treatment for patients with solid malignant tumors. The test gives vital data, including the tumor mutational burden (TMB) score and microsatellite instability (MSI) status, heightening the accuracy and efficiency of treatment plans.
Which Players Are Shaping The Competitive Landscape Of The Oncology Companion Diagnostics Market?
Major companies operating in the oncology companion diagnostics market are F. Hoffmann-La Roche AG, Bayer Diagnostic, Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Laboratory Corporation of America Holdings (LabCorp), Quest Diagnostics Incorporated, Eurofins Scientific SE, Agilent Technologies Inc., Illumina Inc., PerkinElmer Inc., bioMérieux SA, Sysmex Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., Exact Sciences Corporation, BGI Genomics Co. Ltd., Natera Inc., Myriad Genetics Inc., ARUP Laboratories, NeoGenomics Laboratories Inc., Guardant Health, Ambry Genetics Corporation, Biocartis SA, CENTOGENE N.V., Invivoscribe Inc., Pillar Biosciences Inc.
Access The Complete Report Here:
What Are The Emerging Regional Trends Driving The Oncology Companion Diagnostics Market?
North America was the largest region in the oncology companion diagnostic market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the oncology companion diagnostics market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Customize Your Report Here:
https://www.thebusinessresearchcompany.com/customise?id=15623&type=smp
Browse Through More Reports Similar to the Global Oncology Companion Diagnostics Market 2025, By The Business Research Company
Oncology Biosimilar Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/oncology-biosimilar-global-market-report
Interventional Oncology Devices Global Market Report 2025
Clinical Oncology Next Generation Sequencing Global Market Report 2025
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model