Medical Device Clinical Trials Market Outlook 2025–2029: Emerging Opportunities and Key Drivers
Uncover key drivers, emerging technologies, and competitive movements shaping the medical device clinical trials market from 2025–2034 with trusted insights from The Business Research Company
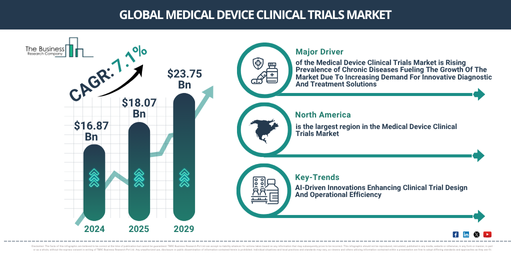
What Is The Present Size Of The Medical Device Clinical Trials Market And What Value Is Projected For 2029?
The medical device clinical trials market size has demonstrated substantial growth in recent years. This market is expected to expand from $16.87 billion in 2024 to $18.07 billion in 2025, achieving a compound annual growth rate (CAGR) of 7.1%. The expansion observed in the historical period stems from various factors, including the increasing prevalence of chronic diseases, a rising demand for innovative medical technologies, regulatory support for clinical research, an expanding aging population, improvements in healthcare infrastructure, and heightened investment from both public and private sectors.
The medical device clinical trials market is projected for substantial expansion over the upcoming years. Its value is anticipated to reach $23.75 billion by 2029, exhibiting a compound annual growth rate (CAGR) of 7.1%. This growth during the forecast period is fueled by factors such as a heightened demand for personalized medical devices, an increased emphasis on real-world evidence, the expanding application of artificial intelligence in trial design and oversight, the proliferation of decentralized and virtual trials, regulatory frameworks facilitating quicker approvals, and a surge in investments aimed at digital health innovations. Key trends foreseen in the same period involve the embrace of decentralized trials, the incorporation of AI and big data analytics, the deployment of wearable monitoring devices, improved regulatory harmonization, and continuous progress in trial design and data analytics.
Access A Free Sample Of The Report:
https://www.thebusinessresearchcompany.com/sample.aspx?id=24295&type=smp
What Factors Are Boosting The Medical Device Clinical Trials Market?
The increasing occurrence of chronic illnesses is anticipated to fuel the expansion of the medical device clinical trials market in the future. These are health conditions that persist over time, develop gradually, and necessitate continuous medical care. Their rise is attributed to inactive lifestyles, where extended periods of sitting or device usage decrease calorie burning and impair cardiovascular well-being, resulting in conditions like obesity, diabetes, and high blood pressure. This surge in chronic diseases creates a demand for medical device clinical trials to create and confirm novel devices for accurate diagnosis, effective treatment, and proper management. An example of this is the report from June 2023 by the Institute for Health Metrics and Evaluation, a US-based public health research institute, stating that over half a billion individuals worldwide had diabetes. This figure is projected to more than double, reaching 1.3 billion people by 2050. Consequently, the escalating occurrence of chronic illnesses is stimulating the growth of the medical device clinical trials market.
What Are The Core Segmentation Criteria In The Medical Device Clinical Trials Market?
The medical device clinical trials market covered in this report is segmented –
1) By Study Type: Feasibility And Pilot Study, Pivotal Study, FDA Premarket Approval (PMA) Application, Post-Approval Study
2) By Indication: Cardiovascular Devices, Neurology Devices, Orthopedic Devices, Diagnostic Imaging, Anesthesia And Respiratory Devices, Other Indications
3) By Study Design: Interventional, Observational, Expanded Access
Subsegments:
1) By Feasibility And Pilot Study: First-in-Human (FIH) Trials, Device Safety Assessment, Procedural Feasibility Studies, Prototype Testing
2) By Pivotal Study: Comparative Effectiveness Studies, Randomized Controlled Trials (RCTs), Non-Inferiority Trials, Superiority Trials
3) By FDA PMA (Pre-Market Approval) Application: Clinical Data Submission, Device Risk Analysis, Effectiveness Evidence Studies, Manufacturing Process Validation
4) By Post-Approval Study: Long-Term Safety Monitoring, Real-World Evidence Collection, Registry Studies, Comparative Outcome Studies
Which Emerging Trends Are Reshaping The Medical Device Clinical Trials Market Landscape?
Major companies engaged in the medical device clinical trials market are concentrating on creating AI-powered medical device software. This endeavor seeks to optimize clinical trial protocols and resource distribution, thereby enhancing trial efficiency and reducing costs. AI-powered medical device software pertains to healthcare applications that harness artificial intelligence (AI) algorithms, especially machine learning or deep learning, to aid or automate medical tasks. For example, in January 2025, Risklick, a pharmaceutical company based in Switzerland, launched Protocol AI. This AI-based software is specifically designed to accelerate and optimize clinical trials for medical devices. Protocol AI utilizes Natural Language Processing (NLP) and Machine Learning (ML) to analyze clinical data, publications, and regulatory documents, automatically generating clinical trial protocols in mere minutes. This innovation considerably decreases the time and expenditure required for protocol development, a process that traditionally takes around six months, and has already shown up to a 50% reduction in document development time for medicinal products.
Which Players Are Shaping The Competitive Landscape Of The Medical Device Clinical TrialsXX Market?
Major companies operating in the medical device clinical trials market are Abbott Laboratories, Siemens Healthineers, Stryker Corporation, Philips Healthcare, Baxter International, Roche Diagnostics, ICON, Intuitive Surgical, Edwards Lifesciences, Fortrea, Medidata, NAMSA, Veranex, TFS HealthScience, Avania, Parexel, Meditrial, Syneos Health, Qserve CRO, Clinius Ltd, Eclevar Medtech, 1med Sa, ISS AG
Download The Full Report Here:
What Regional Trends Are Shaping Growth In The Medical Device Clinical Trials Market?
North America was the largest region in the medical device clinical trials market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the medical device clinical trials market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Get A Report Customized For You Here:
https://www.thebusinessresearchcompany.com/customise?id=24295&type=smp
Browse Through More Reports Similar to the Global Medical Device Clinical Trials Market 2025, By The Business Research Company
Medical Refrigerators Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/medical-refrigerators-global-market-report
Medical Plastics Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/medical-plastics-global-market-report
Medical Adhesive Tapes Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/medical-adhesive-tapes-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model