GMP Cell Therapy Consumables Market Future Outlook: Strong Growth Expected Toward $73.95 Billion by 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the worker heat stress monitoring wearable market from 2026–2035 with trusted insights from The Business Research Company
What is the estimated market size of the GMP Cell Therapy Consumables Market by the end of 2030?
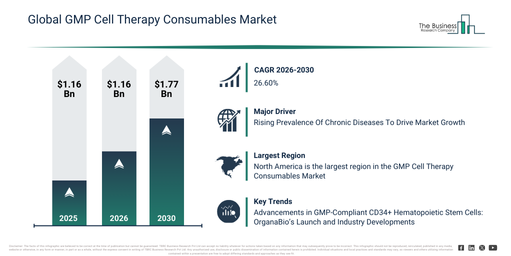
The gmp cell therapy consumables market has experienced substantial expansion in recent years. It is anticipated to increase from $22.73 billion in 2025 to $28.79 billion in 2026, reflecting a compound annual growth rate (CAGR) of 26.7%. This historical growth can be attributed to the proliferation of clinical cell therapy trials, advancements in regenerative medicine research, the growing acceptance of gmp manufacturing standards, heightened investments in cell-based therapies, and the accessibility of sophisticated cell culture instruments.
The gmp cell therapy consumables market is poised for significant expansion in the coming years, with projections indicating it will reach $73.96 billion by 2030, driven by a compound annual growth rate (CAGR) of 26.6%. This anticipated growth stems from factors such as the increasing commercialization of cell therapies, a rising need for scalable GMP production, the expansion of gene and immunotherapy pipelines, a greater focus on process automation, and intensifying regulatory oversight of cell therapy manufacturing. Throughout the forecast period, prominent trends include a heightened demand for GMP-compliant cell therapy consumables, the growing adoption of single-use bioprocessing products, broader utilization of automated cell processing systems, the development of standardized cell culture solutions, and an amplified emphasis on quality and regulatory compliance.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12014&type=smp
What Drivers Are Shaping Future Opportunities In The GMP Cell Therapy Consumables Market?
The growing incidence of chronic diseases is projected to drive the GMP cell therapy consumables market forward. A chronic disease is characterized as an illness lasting three months or more, which can deteriorate over time. The surge in chronic diseases necessitates increased research and development into innovative cell therapies to address these conditions. As cell therapy applications broaden across diverse chronic diseases, the demand for GMP consumables rises, essential for maintaining quality in clinical trials. Rigorous regulatory compliance and patient desire for advanced therapies also contribute significantly to the market’s expansion. For instance, in June 2024, according to the National Health Service, a UK-based government department, 3,615,330 individuals registered with a general practitioner (GP) were diagnosed with non-diabetic hyperglycemia or pre-diabetes (a condition with elevated blood sugar levels, not high enough to be classified as diabetes) in 2023, marking an 18% increase from 3,065,825 cases in 2022. Consequently, the increasing prevalence of chronic diseases is propelling the growth of the GMP cell therapy consumables market.
Which Segments Are Gaining Traction In The GMP Cell Therapy Consumables Market?
The gmp cell therapy consumables market covered in this report is segmented –
1) By Product: Kits, Reagents Or Molecular Biology Reagents, Growth Factors Or Cytokines And Interleukins, Other Products
2) By Cell Therapy: NK Cell Therapy, Stem Cell Therapy, T-Cell Therapy
3) By Process: Cell Collection And Characterization Or Sorting And Separation, Cell Culture And Expansion Or Preparation, Cryopreservation, Cell Processing And Formulation, Cell Isolation And Activation
4) By End-Use: Clinical, Commercial
Subsegments:
1) By Kits: Cell Culture Kits, Cell Isolation Kits, Transfection Kits
2) By Reagents Or Molecular Biology Reagents: Buffers And Solutions, Enzymes, Staining Reagents
3) By Growth Factors Or Cytokines And Interleukins: Hematopoietic Growth Factors, Immunomodulatory Cytokines
4) By Other Products: Cell Culture Vessels, Media And Supplements
How Are New Market Trends Shaping The Landscape Of The GMP Cell Therapy Consumables Market?
Major companies within the GMP cell therapy consumables market are engaged in the development of GMP-compliant CD34+ hematopoietic stem cells (HSCs) to support advanced therapeutic applications. These GMP-compliant CD34+ hematopoietic stem cells (HSCs) are characterized as a distinct type of stem cell that displays the CD34 marker on its surface and is produced under Good Manufacturing Practice (GMP) conditions. Illustratively, in April 2024, OrganaBio, a pharmaceutical company based in the US, introduced HematoPAC-HSC-CB-GMP, providing an on-demand source of GMP-compliant CD34+ hematopoietic stem cells (HSCs) obtained from fresh human cord blood. OrganaBio leverages its considerable experience in cell isolation and GMP manufacturing processes to generate high yields of highly viable CD34+ HSCs within 24 hours of their collection. This product launch is designed to accelerate the creation of next-generation therapies, including those targeting blood cancers and genetic disorders.
Who Are The Top-Performing Companies In The GMP Cell Therapy Consumables Market In Recent Years?
Major companies operating in the gmp cell therapy consumables market are Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, Sartorius AG, Corning Incorporated, Avantor Inc., Fresenius Kabi AG, Miltenyi Biotec, Bio-Techne Corp, STEMCELL Technologies Inc., FUJIFILM Irvine Scientific Inc., BioLife Solutions Inc., PromoCell GmbH, PeproTech Inc., Sino Biological Inc., Wilson Wolf Corporation, Cellexus Ltd., MaxCyte Inc., BioLegend Inc., Repligen Corporation, Terumo Corporation
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/gmp-cell-therapy-consumables-global-market-report
Which Region Currently Holds The Largest Share Of The GMP Cell Therapy Consumables Market?
North America was the largest region in the GMP cell therapy consumables market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the gmp cell therapy consumables market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized GMP Cell Therapy Consumables Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=12014&type=smp
Browse Through More Reports Similar to the Global GMP Cell Therapy Consumables Market 2026, By The Business Research Company
Cell Therapy Technologies Global Market Report 2026
https://www.thebusinessresearchcompany.com/report/cell-therapy-technologies-global-market-report
Cell And Gene Therapy Manufacturing Services Market 2026
Cell And Gene Therapy Clinical Trial Services Global Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company