Closed Viral Vector Manufacturing Platforms Market Research: Growth Projections and Trends 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the closed viral vector manufacturing platforms market from 2026–2035 with trusted insights from The Business Research Company
How is the Closed Viral Vector Manufacturing Platforms Market size predicted to change over the forecast window of 2026–2035?
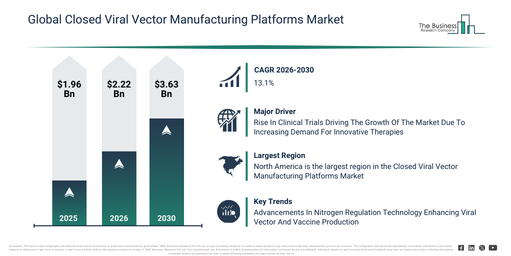
The market size for closed viral vector manufacturing platforms has experienced rapid growth in recent years. This market is expected to expand from $1.96 billion in 2025 to $2.22 billion in 2026, achieving a compound annual growth rate (CAGR) of 13.4%. Historically, this growth can be ascribed to a surge in gene therapy clinical trials, the increased deployment of viral vectors in vaccine development, the broadening of cell and gene therapy research programs, a heightened regulatory focus on sterile manufacturing, and the early adoption of closed processing technologies.
The closed viral vector manufacturing platforms market size is projected to experience substantial expansion in the coming years. This market is anticipated to reach $3.63 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 13.1%. Factors contributing to this growth during the forecast timeframe include the accelerating commercialization of gene therapies, an escalating need for extensive viral vector supplies, increasing capital expenditure in cutting-edge biomanufacturing facilities, the broadening of contract manufacturing capabilities, and a heightened emphasis on standardizing and automating processes. Key trends expected over the forecast period encompass a greater uptake of entirely closed and automated production systems, a surging requirement for scalable viral vector manufacturing platforms, the wider incorporation of single-use and modular parts, the broadening of complete closed processing workflows, and an intensified focus on adherence to regulations and managing contamination.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28981&type=smp
Which Drivers Are Contributing To The Expansion Of The Closed Viral Vector Manufacturing Platforms Market?
An increasing volume of clinical trials is anticipated to drive expansion in the closed viral vector manufacturing platforms market moving ahead. These trials involve meticulously planned human studies aimed at evaluating the safety, efficacy, and suitability of novel medical treatments or procedures. The escalating need for inventive therapies fuels the demand for clinical trials, as the rising incidence of chronic and intricate diseases necessitates the creation and evaluation of new drugs and treatments to meet currently unaddressed medical requirements. Closed viral vector manufacturing platforms facilitate clinical trials through the reliable and aseptic production of viral vectors, thereby guaranteeing the safety and dependability of the therapeutic agents being studied. As an illustration, data from December 2024, provided by the Association of the British Pharmaceutical Industry (ABPI), a trade association based in the UK, indicated that the aggregate count of industry-funded clinical trials initiated in the UK climbed from 411 in 2022 to 426 in 2023. Consequently, the increasing volume of clinical trials is a key impetus for the expansion of the closed viral vector manufacturing platforms market.
What Segment Categories Are Covered In The Closed Viral Vector Manufacturing Platforms Market?
The closed viral vector manufacturing platforms market covered in this report is segmented –
1) By Product Type: Plasmid Deoxyribonucleic Acid (DNA), Lentiviral Vectors, Adenoviral Vectors, Adeno-Associated Viral Vectors, Retroviral Vectors, Other Product Types
2) By Workflow: Upstream Processing, Downstream Processing, Fill-Finish
3) By Application: Gene Therapy, Vaccines, Cell Therapy, Other Applications
4) By End User: Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Manufacturing Organizations, Other End Users
Subsegments:
1) By Plasmid Deoxyribonucleic Acid (DNA): Supercoiled DNA, Linearized DNA, Minicircle DNA
2) By Lentiviral Vectors: Integrating Lentiviral Vectors, Non-Integrating Lentiviral Vectors
3) By Adenoviral Vectors: Replication-Deficient Adenoviral Vectors, Replication-Competent Adenoviral Vectors
4) By Adeno-Associated Viral Vectors (AAV): Single-Stranded AAV (ssAAV), Self-Complementary AAV (scAAV)
5) By Retroviral Vectors: Gamma-Retroviral Vectors, Alpha-Retroviral Vectors
6) By Other Product Types: Herpes Simplex Virus (HSV) Vectors, Vaccinia Virus Vectors, Baculovirus Vectors
Which Trends Are Shaping Activity Within The Closed Viral Vector Manufacturing Platforms Market?
Leading firms within the closed viral vector manufacturing platforms market are concentrating on creating sophisticated solutions, like nitro controllers, aimed at averting nitrogen-induced stress or toxicity in cell cultures. These nitro controllers function as devices or systems designed to monitor and regulate nitrogen levels or reactive nitrogen species during bioprocesses, thereby ensuring ideal conditions for cell growth and product generation. An example of this is Univercells Technologies, a biotechnology company from Belgium, which introduced the scale-X nitro controller in May 2025. This system is both compact and cost-effective, engineered specifically for large-scale production of viral vectors and vaccines. It offers compatibility with 200 m² and 600 m² scale-X nitro bioreactors and can accommodate both adherent and suspension cell cultures. The system facilitates the efficient production of AAV, lentiviral vectors, exosomes, and oncolytic viruses. Furthermore, it permits swift scale-up, streamlining the progression from research and development phases to commercial manufacturing within an eight-month timeframe.
Which Leading Companies Dominate The Closed Viral Vector Manufacturing Platforms Market Share?
Major companies operating in the closed viral vector manufacturing platforms market are Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, WuXi AppTec, Samsung Biologics Co. Ltd., FUJIFILM Biotechnologies, Oxford Biomedica Plc, Hillgene, Takara Bio Inc., SkyPharma Production SAS, GeneScript ProBio, Obio Technology Corp. Ltd., VectorBuilder, Charles River Laboratories Pvt. Ltd., Creative Biogene, Esco Aster Pte. Ltd., Karma Biotechnologies, Cell and Gene Therapy Catapult, CoJourney, Applied Biological Laboratories Inc., CEVEC Pharmaceuticals, Genesail Biotech Co. Ltd.
Access The Complete Report For Deeper Market Insights:
How Does The Closed Viral Vector Manufacturing Platforms Market Perform Across Major Global Regions?
North America was the largest region in the Closed Viral Vector Manufacturing Platforms market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the closed viral vector manufacturing platforms market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Closed Viral Vector Manufacturing Platforms Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=28981&type=smp
Browse Through More Reports Similar to the Global Closed Viral Vector Manufacturing Platforms Market 2026, By The Business Research Company
Viral Vector Manufacturing Market Report 2026
https://www.thebusinessresearchcompany.com/report/viral-vector-manufacturing-global-market-report
Viral And Non Viral Vector Manufacturing Market Report 2026
Oral Expectorant Market Report 2026
https://www.thebusinessresearchcompany.com/report/oral-expectorant-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model