Pharmaceutical Quality Control Market Forecast: How Big Will It Be by 2030?
Uncover key drivers, emerging technologies, and competitive movements shaping the pharmaceutical quality control market from 2026–2035 with trusted insights from The Business Research Company
What level of growth is expected in the Pharmaceutical Quality Control Market between 2026 and 2030?
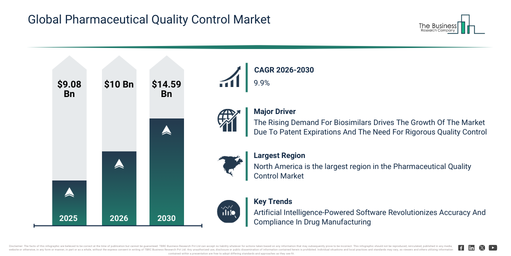
The pharmaceutical quality control market size has witnessed rapid expansion in recent years. This market is anticipated to expand from $9.08 billion in 2025 to $10 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 10.2%. The growth experienced in the past can be attributed to limited access to advanced qc instruments, reliance on manual testing methods, growing pharmaceutical manufacturing, increasing regulatory compliance requirements, and expansion of microbiological testing capabilities.
The pharmaceutical quality control market is anticipated to experience robust growth in the upcoming years, with its size projected to reach $14.59 billion by 2030, growing at a compound annual growth rate (CAGR) of 9.9%. This expansion during the forecast period is attributed to several factors, including the integration of cloud-based and AI-enabled QC solutions, the increasing adoption of automated analytical instruments, rising investment in validation and qualification services, a growing emphasis on sterility assurance and stability testing, and the broadening of contract research and development partnerships. Major trends foreseen for the forecast period involve the widespread adoption of advanced chromatography and spectroscopy instruments, an increasing demand for microbiological and sterility testing services, the expansion of stability testing and validation services, a heightened focus on endotoxin and bioburden analysis, and the growing requirement for both on-premises and cloud-based quality control solutions.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=27380&type=smp
Which Drivers Are Expected To Impact The Pharmaceutical Quality Control Market During The Forecast Period?
The growing need for biosimilars is projected to boost the expansion of the pharmaceutical quality control market in the future. Biosimilars are defined as biological medical products that are highly comparable to an already approved reference biologic, demonstrating no clinically significant distinctions in their safety, purity, and potency. The heightened demand for biosimilars primarily originates from the expiration of patents on original biologic drugs, which facilitates the entry of more cost-effective alternatives into the market. Pharmaceutical quality control ensures the safety, purity, and consistency of biosimilars by rigorously testing them against reference biologics, thereby supporting regulatory compliance and market approval. For instance, in January 2023, Cardinal Health Inc., a US-based healthcare company, reported that as of January 2022, there were 33 FDA-approved biosimilar products in the US, with 21 available on the market. By 2023, this figure had risen to 40 FDA-approved biosimilars, with 25 commercially available. Consequently, the increasing demand for biosimilars is a key driver for the growth of the pharmaceutical quality control market.
Which Segments Are Driving Activity In The Pharmaceutical Quality Control Market?
The pharmaceutical quality control market covered in this report is segmented –
1) By Product: Consumables, Services, Instruments
2) By Deployment Type: Cloud-Based And Web-Based, On-Premises
3) By Analysis Type: Sterility Testing, Membrane Filtration, Direct Inoculation, Bio Burden Testing, Aerobic Count Testing, Anaerobic Count Testing, Spore Count Testing, Fungi or Mold Count Testing, Endotoxin Testing, Other Analysis Types
4) By End-user: Pharmaceutical And Biotechnology Companies, Contract Development and Manufacturing Organization Or Contract Research Organization, Research Laboratories
Subsegments:
1) By Consumables: Culture Media, Reagents, Assay Kits, Filters And Membranes, Pipette Tips
2) By Services: Microbiological Testing Services, Analytical Testing Services, Stability Testing Services, Sterility Assurance Services, Validation And Qualification Services
3) By Instruments: Chromatography Systems, Spectroscopy Instruments, Particle Size Analyzers, Potential Of Hydrogen Meters And Electrical Conductivity Meters, Autoclaves And Incubators
Which Trends Are Influencing The Development Of The Pharmaceutical Quality Control Market?
Companies leading the pharmaceutical quality control market are prioritizing technological advancements, such as intelligent software platforms, to improve testing precision and regulatory adherence. Intelligent software platforms in pharmaceutical quality control are sophisticated digital systems that integrate automation, data analytics, and artificial intelligence (AI) to enhance the accuracy, efficiency, and compliance of testing procedures. As an illustration, in June 2025, Dycine Pharmaceuticals Ltd., an India-based pharmaceutical company, introduced an artificial intelligence-driven quality control platform. This platform was developed to elevate manufacturing standards and guarantee product integrity. It leverages advanced artificial intelligence algorithms to identify anomalies, monitor real-time data, and streamline compliance processes throughout the pharmaceutical production lifecycle. The system provides key capabilities like predictive analytics, automated reporting, and seamless integration with existing manufacturing systems, thereby helping to minimize errors, boost efficiency, and uphold consistent quality across all production batches.
Which Major Firms Are Strengthening Their Position In The Pharmaceutical Quality Control Market?
Major companies operating in the pharmaceutical quality control market are Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Merck KGaA, Becton Dickinson and Company, SGS S.A., Eurofins Scientific SE, WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., PerkinElmer Inc., Mettler-Toledo International Inc., bioMérieux SA, Shimadzu Scientific Instruments Inc., Waters Corporation, Randox Laboratories Ltd., SOLVIAS AG, Esco Micro Pte. Ltd, REMI Elektrotechnik Limited, Lucideon Limited, BRAM-COR S.p.A.
Access The Complete Report For Deeper Market Insights:
Which Region Shows The Strongest Potential For Future Expansion In The Pharmaceutical Quality Control Market?
North America was the largest region in the pharmaceutical quality control market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the pharmaceutical quality control market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Pharmaceutical Quality Control Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=27380&type=smp
Browse Through More Reports Similar to the Global Pharmaceutical Quality Control Market 2026, By The Business Research Company
Handheld Laser Meter Market Report 2026
https://www.thebusinessresearchcompany.com/report/handheld-laser-meter-global-market-report
Road Profile Laser Sensor Market Report 2026
https://www.thebusinessresearchcompany.com/report/road-profile-laser-sensor-global-market-report
Laser Marking Machine Market 2026
https://www.thebusinessresearchcompany.com/report/laser-marking-machine-market
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model