Pharmaceutical Stability And Storage Services Market Dynamics 2026: Future Outlook and Key Insights
Uncover key drivers, emerging technologies, and competitive movements shaping the pharmaceutical stability and storage services market from 2026–2035 with trusted insights from The Business Research Company
What size range is anticipated for the Pharmaceutical Stability And Storage Services Market from 2026 to 2030?
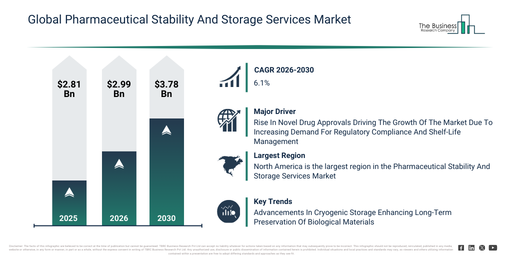
The pharmaceutical stability and storage services market has demonstrated robust growth over recent years. This market is set to expand from $2.81 billion in 2025 to $2.99 billion in 2026, achieving a compound annual growth rate (CAGR) of 6.1%. Historically, its expansion has been influenced by factors including stringent pharmaceutical regulatory requirements, an increase in global drug production, the expansion of biologics manufacturing, the demand for shelf-life validation, and rising clinical trial volumes.
The pharmaceutical stability and storage services market is projected to experience robust expansion in the coming years, reaching $3.78 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 6.1%. This anticipated growth is primarily driven by factors such as the increasing development of biologics and cell therapies, a rise in pharmaceutical outsourcing, the ongoing expansion of global cold chain infrastructure, an uptick in vaccine production, and more stringent regulatory oversight on drug stability. Key trends expected during this forecast period encompass a growing demand for regulatory-compliant storage solutions, an increase in the outsourcing of stability testing services, an expanding requirement for cold chain and cryogenic storage, the broadening of stability services tailored for biologics, and a heightened emphasis on ensuring long-term drug quality.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=23473&type=smp
What Drivers Are Affecting Demand In The Pharmaceutical Stability And Storage Services Market?
A rising volume of novel drug authorizations is anticipated to boost the expansion of the pharmaceutical stability and storage services market in the future. Specifically, a novel drug is defined as a recently developed pharmaceutical compound that has not received prior approval for use or marketing. This uptick in novel drug approvals stems from progress in biotechnology, which allows for the identification of new compounds, coupled with enhanced regulatory frameworks that streamline drug development, making it quicker and more effective. The growing incidence of novel drug approvals fuels the need for pharmaceutical stability and storage services, which are crucial for maintaining optimal conditions, ensuring regulatory compliance, and managing extended shelf-life. As an illustration, in October 2023, the U.S. Food and Drug Administration (FDA), a US-based federal agency responsible for protecting public health, reported that 37 novel drugs were approved in 2022, with an additional 50 in 2024. These approvals are advancing treatments for conditions such as cancer, rare diseases, neurology, infections, and other ailments including schizophrenia, pain, and Alzheimer’s. Consequently, the continued rise in novel drug approvals serves as a significant impetus for the growth of the pharmaceutical stability and storage services market.
Which Segment Groups Are Influencing The Pharmaceutical Stability And Storage Services Market?
The pharmaceutical stability and storage services market covered in this report is segmented –
1) By Service: Stability, Storage
2) By Product Type: Tablets, Capsules, Injectable Solutions, Oral Suspensions, Combination Products, Other Product Types
3) By Molecule: Small Molecule, Large Molecule
4) By Mode: In-House, Outsourcing
5) By End User: Biopharmaceutical Companies, Contract Manufacturing Organizations, Contract Research Organizations, Other End Users
Subsegments:
1) By Stability Services: Long-Term Stability Studies, Accelerated Stability Studies, Intermediate Stability Studies, Photostability Testing, Forced Degradation Studies
2) By Storage Services: Ambient Storage, Refrigerated Storage (2°C to 8°C), Frozen Storage (-20°C to -80°C), Cryogenic Storage (-150°C to -196°C), Controlled Drug Substance And Product Storage
What Trends Are Driving The Growth Trajectory Of The Pharmaceutical Stability And Storage Services Market?
Companies operating within the pharmaceutical stability and storage services market are increasingly focusing on developing sophisticated solutions, such as cryogenic storage suites, to improve the preservation of temperature-sensitive pharmaceutical products. Cryogenic storage suites are specialized facilities designed to maintain extremely low temperatures, typically below -150°C, which is crucial for preserving delicate biological and pharmaceutical materials like cell therapies, vaccines, and biologics. An illustrative example occurred in December 2024 when Astoriom, a UK-based company in the stability and biospecimen storage sector, inaugurated a cryogenic storage suite at its Tramore facility in County Waterford, Ireland. This launch represents a significant expansion of its capabilities for biological sample storage. This cutting-edge suite is engineered to meet the rising demand for the secure, long-term preservation of biological materials, thereby ensuring their viability over extended periods. The storage offers precise temperature management down to -196°C, substantial storage capacity, ISO 9001 certification for regulatory compliance, and advanced security features, including access controls and monitoring systems.
Who Are The Top-Performing Companies In The Pharmaceutical Stability And Storage Services Market In Recent Years?
Major companies operating in the pharmaceutical stability and storage services market are SGS S.A., Eurofins Scientific SE, Intertek Group plc, Catalent Inc., Almac Group, Alcami Corporation, Reading Scientific Services Ltd., Charles River Laboratories, Element Materials Technology Inc., Precision Stability Storage, Roylance Stability Storage Limited, BioLife Solutions Inc., Alloga UK Ltd., Auriga Research Private Limited, Q Laboratories Inc., ALS Limited, Lucideon Limited, Cencora Inc., PCI Pharma Services, DHL Life Sciences & Healthcare
Access The Complete Report For Deeper Market Insights:
Which Region Currently Holds The Largest Share Of The Pharmaceutical Stability And Storage Services Market?
North America was the largest region in the pharmaceutical stability and storage services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the pharmaceutical stability and storage services market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Pharmaceutical Stability And Storage Services Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=23473&type=smp
Browse Through More Reports Similar to the Global Pharmaceutical Stability And Storage Services Market 2026, By The Business Research Company
Pharmacy Repackaging Systems Market Report 2026
https://www.thebusinessresearchcompany.com/report/pharmacy-repackaging-systems-global-market-report
Temperature Controlled Pharmaceutical Containers Market Report 2026
Pharmaceutical Packaging Equipment Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model