Sterile Injectables CDMO Market Outlook: Growth and Forecast Highlights 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the sterile injectables cdmo market from 2026–2035 with trusted insights from The Business Research Company
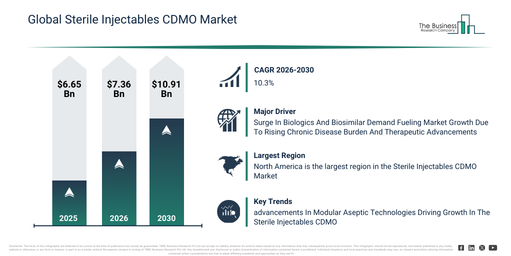
What is the expected shift in the Sterile Injectables CDMO Market’s size between 2026 and 2030?
The sterile injectables CDMO market size has seen swift expansion over recent years. It is anticipated to expand from $6.65 billion in 2025 to $7.36 billion in 2026, achieving a compound annual growth rate (CAGR) of 10.7%. The historical market growth is attributable to factors including restricted CDMO infrastructure for sterile injectables, an increasing need for small molecule injectables, a reliance on in-house formulation development, intensifying regulatory oversight for sterile manufacturing, and the surge in global pharmaceutical output.
The sterile injectables cdmo market is projected to experience swift expansion in the coming years, anticipated to reach a valuation of $10.91 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 10.3%. This growth during the forecast period can be attributed to several factors, including the increased manufacturing of large molecule biologics, a surge in demand for cell and gene therapies, the implementation of sophisticated aseptic processing technologies, a greater reliance on outsourcing to cdmos, and an escalation in the need for specialized packaging and cold chain storage. Key trends anticipated in the forecast period encompass an escalating demand for contract manufacturing services for sterile injectables, an expansion of analytical and testing capabilities to ensure quality, an increase in the production of monoclonal antibodies, peptides, and cell/gene therapies, a growing preference for lyophilized, emulsion, and reconstitutable injectable formulations, and the integration of formulation development with comprehensive packaging and storage solutions.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25912&type=smp
Which Drivers Are Shaping Strategic Decisions In The Sterile Injectables CDMO Market?
The future expansion of the sterile injectables CDMO market is anticipated to be driven by the escalating need for biologics and biosimilars. Biologics are pharmaceuticals created from living cells or organisms that precisely target disease components, whereas biosimilars are almost identical versions of approved biologics, offering comparable quality, safety, and efficacy. This heightened demand for such innovative therapies stems from a growing prevalence of chronic and intricate health conditions, as they provide more precise and potent treatment options than conventional drugs. Consequently, the expanding use of biologics and biosimilars elevates the reliance on sterile injectable CDMO services, since these treatments are typically administered through injections and necessitate rigorous sterile environments to guarantee patient safety, product quality, and compliance with regulations. An illustration of this trend is found in the US, where, as reported by the Center for Biosimilars, a US-based nonprofit research organization, in February 2025, an estimated US $324 billion was expended on biologic medicines in 2023, constituting approximately 45% of the country’s total prescription drug spending. Thus, the expanding requirement for biologics and biosimilars is undeniably propelling the growth of the sterile injectables CDMO market.
What Segment Classifications Make Up The Sterile Injectables CDMO Market?
The sterile injectables cdmo market covered in this report is segmented –
1) By Type: Small Molecule, Large Molecule
2) By Service: Formulation Development, Analytical And Testing Services, Manufacturing, Packaging, Storage, Other Services
3) By Route Of Administration: Subcutaneous (SC), Intravenous (IV), Intramuscular (IM), Other Routes Of Administration
4) By Therapeutic Area: Oncology, Cardiovascular Diseases, Central Nervous System Diseases, Infectious Disorders, Musculoskeletal Diseases, Hormonal Diseases, Other Therapeutic Areas
5) By End-User: Pharmaceutical Companies, Biopharmaceutical Companies, Research Institutes, Other End-Users
Subsegments:
1) By Small Molecule: Solutions, Suspensions, Lyophilized Injectables, Emulsions, Reconstitutable Powders
2) By Large Molecule: Monoclonal Antibodies (mAbs), Peptides And Proteins, Vaccines, Cell And Gene Therapies, Insulins And Biosimilars
What Upcoming Trends Are Likely To Define The Future Path Of The Sterile Injectables CDMO Market?
Leading companies in the Sterile Injectables CDMO market are concentrating on creating advanced solutions, such as modular aseptic filling platforms, to boost flexibility and efficiency in sterile drug production. Modular aseptic filling platforms are adaptable isolator-based sterile filling systems built with interchangeable modules, enabling CDMOs to quickly transition between vial, syringe, or cartridge formats. This helps reduce changeover time, minimize product loss, and effectively support small- to mid-scale batches for both clinical and commercial sterile injectables. As an illustration, in February 2025, Recipharm AB, a Sweden-based contract development and manufacturing organization (CDMO), launched a fully operational modular sterile filling system at its Wasserburg, Germany site. This system is designed for aseptic filling of small to mid-scale batches (500–50,000 units), minimizes product loss (less than 500 milliliters), supports interchangeable filling modules, and facilitates efficient processing of high-value biologics within a completely enclosed isolator.
Which Major Industry Participants Are Leading The Sterile Injectables CDMO Market Growth?
Major companies operating in the sterile injectables cdmo market are Pfizer Inc., Thermo Fisher Scientific Inc., Boehringer Ingelheim, Fresenius Kabi AG, Lonza Group Ltd., Catalent Inc., Samsung Biologics Co. Ltd., Fareva SA, Recipharm AB, PCI Pharma Services, Vetter Pharma, Delpharm, Kindeva Drug Delivery LP, Ajinomoto Bio-Pharma Services, Cenexi, Grand River Aseptic Manufacturing Inc., INCOG BioPharma Services, Aurigene Pharmaceutical Services, Jubilant HollisterStier LLC, Symbiosis Pharmaceutical Services Ltd., Afton Scientific Corp., Amaran Biotechnology Inc.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/sterile-injectables-cdmo-global-market-report
Which Region Is Expected To Experience The Fastest Growth In The Sterile Injectables CDMO Market?
North America was the largest region in the sterile injectables CDMO market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the sterile injectables cdmo market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Sterile Injectables CDMO Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=25912&type=smp
Browse Through More Reports Similar to the Global Sterile Injectables CDMO Market 2026, By The Business Research Company
Generic Oncology Sterile Injectable Market Report 2026
Injectable Drug Delivery Devices Market Report 2026
Sterile Medical Packaging Market Report 2026
https://www.thebusinessresearchcompany.com/report/sterile-medical-packaging-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model