Global Chlamydia Infection Diagnostics And Therapeutics Market Set for Strong Expansion, Reaching $1.98 Billion With 5.6% CAGR by 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the chlamydia infection diagnostics and therapeutics market from 2026–2035 with trusted insights from The Business Research Company
What long-term size projections exist for the Chlamydia Infection Diagnostics And Therapeutics Market covering 2026–2035?
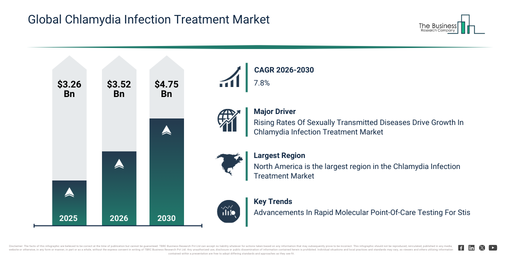
The chlamydia infection diagnostics and therapeutics market has experienced substantial growth in recent years. It is projected to increase from $1.51 billion in 2025 to $1.59 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 5.8%. The expansion during the historic period can be attributed to the rising prevalence of sexually transmitted infections, the broadening of public health screening initiatives, the availability of effective antibiotic therapies, growing awareness of reproductive health, and advancements in diagnostic laboratory infrastructure.
The chlamydia infection diagnostics and therapeutics market is anticipated to experience substantial growth in the upcoming years, with projections indicating an expansion to $1.98 billion by 2030 at a compound annual growth rate (CAGR) of 5.6%. This growth during the forecast period is attributed to several factors, including the increasing adoption of advanced NAAT technologies, rising investments in sexual health programs, the proliferation of home-based diagnostic solutions, a growing emphasis on antimicrobial stewardship, and an increasing demand for integrated diagnostic-therapeutic platforms. Major trends observed in this period include the rising adoption of molecular diagnostic testing, an increase in the use of rapid point-of-care diagnostics, a growing preference for combined diagnostic and therapeutic approaches, the expansion of antibiotic resistance monitoring, and an enhanced focus on early detection and screening programs.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=18295&type=smp
Which Drivers Are Impacting The Competitive Landscape Of The Chlamydia Infection Diagnostics And Therapeutics Market?
The increasing occurrence of sexually transmitted infections is projected to drive expansion within the chlamydia infection diagnostics and therapeutics market in the future. These infections are defined as conditions commonly spread via sexual intercourse, encompassing vaginal, anal, or oral contact. The surge in STIs stems from factors such as inadequate or incorrect use of barrier contraception, insufficient knowledge regarding safe sexual behaviors, expanded global travel and migration, and a potential reduction in public health funding for preventative initiatives. Early detection and efficient treatment, facilitated by chlamydia infection diagnostics and therapeutics, are crucial for effective management of sexually transmitted infections to avert complications and further spread. As an illustration, the Minority HIV/AIDS Fund (MHAF), a US-based government agency, reported in October 2025 that an estimated 39.9 million individuals globally were living with HIV in 2023, comprising 38.6 million adults and 1.4 million children, which represents an increase from prior years. Consequently, the escalating global prevalence of sexually transmitted infections is propelling the growth of the chlamydia infection diagnostics and therapeutics market.
What Leading Segments Are Studied In The Chlamydia Infection Diagnostics And Therapeutics Market?
The chlamydia infection diagnostics and therapeutics market covered in this report is segmented –
1) By Type: Nucleic Acid Amplification Tests (NAATS), Direct Fluorescent Tests, Macrolides, Quinolones, Sulfonamides, Tetracycline, Aminopenicillins
2) By Type Of Infections: Genital Chlamydia Infection, Rectal Chlamydia Infection, Ocular Chlamydia Infection
3) By Application: Hospitals, Specialty Clinics, Diagnostic Centers Therapeutics, Hospital Pharmacies, Drugstores, Retail Pharmacies, Online Pharmacies
Subsegments:
1) By Nucleic Acid Amplification Tests (NAATS): PCR-Based Tests, LAMP-Based Tests
2) By Direct Fluorescent Tests: Direct Fluorescent Antibody Staining, Direct Fluorescent In-Situ Hybridization
3) By Macrolides: Azithromycin, Erythromycin
4) By Quinolones: Ciprofloxacin, Ofloxacin
5) By Sulfonamides: Sulfamethoxazole, Trimethoprim-Sulfamethoxazole
6) By Tetracycline: Doxycycline, Tetracycline
7) By Aminopenicillins: Amoxicillin, Ampicillin
What Trends Are Projected To Support The Growth Of The Chlamydia Infection Diagnostics And Therapeutics Market?
Prominent companies operating within the chlamydia market are prioritizing the development of innovative solutions, such as the growth of Point-of-Care Molecular Diagnostics, to achieve faster, more accurate onsite detection and improve timely treatment outcomes. Point-of-care molecular diagnostics expansion refers to the increased utilization of rapid, onsite molecular tests that enable immediate detection of infections like chlamydia without reliance on centralized laboratories. For instance, in January 2025, F. Hoffmann-La Roche AG, a Switzerland-based healthcare company, secured FDA 510(k) clearance alongside a CLIA waiver for its cobas liat molecular tests, specifically designed for diagnosing sexually transmitted infections (STIs) directly at the point of care. These tests incorporate multiplex assay panels capable of detecting and differentiating various STIs, including chlamydia, gonorrhea, and Mycoplasma genitalium, from a single patient sample, providing results in 20 minutes or less.
Which Leading Companies Dominate The Chlamydia Infection Diagnostics And Therapeutics Market Share?
Major companies operating in the chlamydia infection diagnostics and therapeutics market are F. Hoffman La-Roche Ltd, Merck & Co. Inc., AbbVie Inc, Sanofi S.A., Bristol-Myers Squibb, AstraZeneca, Abbott Laboratory, Novartis AG, GlaxoSmithKline plc, Takeda Pharmaceutical, Danaher Corporation, Gilead Sciences, Siemens Healthineers, Becton Dickinson and Company, Accelerate Diagnostics, Hologic Inc., Biomerieux, QuidelOrtho Corporation, Bio-Rad Laboratories Inc., DiaSorin SpA, Luminex Corporation, Meridian Bioscience, Trinity Biotech Plc, Copan Diagnostics, Hain Lifescience GmbH
Access The Complete Report For Deeper Market Insights:
Which Region Is Anticipated To See The Fastest Growth In The Chlamydia Infection Diagnostics And Therapeutics Market?
North America was the largest region in the chlamydia infection diagnostics and therapeutics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the chlamydia infection diagnostics and therapeutics market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Chlamydia Infection Diagnostics And Therapeutics Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=18295&type=smp
Browse Through More Reports Similar to the Global Chlamydia Infection Diagnostics And Therapeutics Market 2026, By The Business Research Company
Chlamydia Trachomatis Ct And Neisseria Gonorrhoeae Ng Testing Market Report 2026
Chlamydia Infection Diagnostics Market Report 2026
Chlamydia Infection Treatment Market Report 2026
https://www.thebusinessresearchcompany.com/report/chlamydia-infection-treatment-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model