Endotoxin Testing Market Future Outlook: Strong Growth Expected Toward $3.11 Billion by 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the endotoxin testing market from 2026–2035 with trusted insights from The Business Research Company
What is the estimated market size of the Endotoxin Testing Market by the end of 2030?
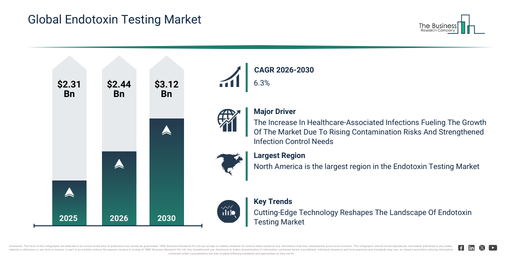
The endotoxin testing market has experienced robust expansion in its size over the past few years. Projections indicate it is set to increase from $2.31 billion in 2025 to $2.44 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 5.5%. This historical growth is attributable to factors such as drug safety regulations, an increase in pharmaceutical manufacturing, adherence to medical device quality standards, heightened patient safety concerns, and the broadening of laboratory testing capabilities.
The endotoxin testing market size is anticipated to undergo substantial growth in the upcoming years. It is projected to expand to $3.12 billion by 2030, achieving a compound annual growth rate (CAGR) of 6.3%. This projected growth can be attributed to several factors including the expansion of biologic drugs, stricter regulatory audits, increasing automation in quality testing processes, continued innovation in medical devices, and enhanced contamination risk management strategies. Furthermore, key trends for the forecast period include an elevated focus on pharmaceutical safety, more rigorous regulatory compliance requirements, a rise in medical device quality testing, the broadening of in vitro diagnostic testing applications, and a persistent demand for accurate endotoxin detection.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12932&type=smp
What Drivers Are Shaping Future Opportunities In The Endotoxin Testing Market?
The anticipated expansion of the endotoxin testing market is linked to the rising occurrence of healthcare-associated infections (HAIs). HAIs are defined as infections contracted during medical treatment that were absent upon a patient’s admission to a hospital. Factors contributing to the increase in HAIs include insufficient infection prevention and control measures, contact with tainted medical equipment, and the growing spread of drug-resistant pathogens. Patient safety is improved through endotoxin testing, which identifies bacterial endotoxins in medical equipment, injectable pharmaceuticals, and implantable devices, consequently lowering the likelihood of sepsis, inflammation, and complications linked to devices. For example, in November 2024, the World Health Organization (WHO), a public health agency based in Switzerland, stated in its Global Report on Infection Prevention and Control 2024 that despite 71% of countries having active IPC programmes, only 6% fulfilled all WHO minimum IPC requirements between 2023–2024, thus exposing millions of patients to an elevated risk of HAIs. Consequently, the expanding occurrence of healthcare-associated infections is fueling the growth of the endotoxin testing market.
Which Segments Are Gaining Traction In The Endotoxin Testing Market?
The endotoxin testing market covered in this report is segmented –
1) By Test Type: LAL (Limulus Amebocyte Lysate) Test, Chromogenic Tests, Turbidimetric Tests, Gel Clot Tests, MAT Test, Rabbit Pyrogen Test, Recombinant Factor C (rFC) Assay
2) By Application: Medical Devices, Pharmaceuticals, Packaging, Raw Materials
3) By End-User: Hospitals, Laboratories, Research Institutes
Subsegments:
1) By LAL (Limulus Amebocyte Lysate) Test: Gel Clot LAL Test, Kinetic LAL Test, Endpoint LAL Test
2) By Chromogenic Tests: Chromogenic LAL Test, Chromogenic Factor C Assay
3) By Turbidimetric Tests: Turbidimetric LAL Test, Turbidimetric Factor C Assay
4) By Gel Clot Tests: Gel Clot LAL Test
5) By MAT Test (Monocyte Activation Test): Direct MAT, Indirect MAT
6) By Rabbit Pyrogen Test: Standard Rabbit Pyrogen Test
7) By Recombinant Factor C (rFC) Assay: RFC Assay For Endotoxin Detection
Which Trends Are Guiding The Evolution Of The Endotoxin Testing Market?
Leading companies in the endotoxin testing market are concentrating on developing sophisticated solutions, such as integrated absorbance microplate readers, to enhance testing accuracy, streamline laboratory workflows, and reinforce compliance with data integrity requirements. Advanced absorbance readers in endotoxin testing are instruments crafted for seamless integration with specialized assay software, supporting validated workflows, and delivering consistent results aligned with industry standards. For instance, in August 2023, Lonza Group AG, a Switzerland-based pharmaceutical, biotechnology, and nutrition manufacturing company, introduced the Nebula Absorbance Reader, a next-generation microplate reader designed to simplify endotoxin and pyrogen testing. Fully integrated with Lonza’s latest WinKQCL software, this system facilitates data integrity compliance, reduces the validation burden, and minimizes training needs by allowing existing WinKQCL users to operate the new reader without needing to learn additional software. Developed as a technologically advanced substitute for the discontinued ELx808 absorbance reader, the Nebula platform provides comparable performance and is optimized for Lonza’s absorbance-based endotoxin assays, including the PYROGENT 5000 Turbidimetric and Kinetic-QCL Chromogenic Endotoxin Assays.
Who Are The Top-Performing Companies In The Endotoxin Testing Market In Recent Years?
Major companies operating in the endotoxin testing market are Thermo Fisher Scientific Inc., Merck KGaA, bioMérieux SA., Eurofins Scientific SE, Lonza Group Ltd., WuXi AppTec Co. Ltd., Bio-Rad Laboratories Inc., Maravai LifeSciences Holdings Inc., Cambrex Corporation, GenScript Biotech Corporation, Charles River Laboratories International Inc., Nelson Laboratories LLC, Lifecore Biomedical Inc., Stellar Biotechnologies Inc., Biovision Inc., Associates of Cape Cod Inc., FUJIFILM Wako Pure Chemical Corporation, InvivoGen Inc., Tebu-bio Nv., MatTek Corporation, Microcoat Biotechnologie GmbH, Accelagen Inc., BioAssay Systems LLC, BioThema AB, Hycult Biotech Inc., Pacific Biolabs Inc., Xiamen Bioendo Technology Co. Ltd., Zhanjiang A&C Biological Ltd., Accugen Laboratories Inc.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/endotoxin-testing-global-market-report
Which Region Is Anticipated To See The Fastest Growth In The Endotoxin Testing Market?
North America was the largest region in the endotoxin testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the endotoxin testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Endotoxin Testing Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=12932&type=smp
Browse Through More Reports Similar to the Global Endotoxin Testing Market 2026, By The Business Research Company
In Vitro Toxicology Testing Market Report 2026
https://www.thebusinessresearchcompany.com/report/in-vitro-toxicology-testing-global-market-report
Immunotoxin Market Report 2026
https://www.thebusinessresearchcompany.com/report/immunotoxin-global-market-report
Genetic Toxicology Testing Market Report 2026
https://www.thebusinessresearchcompany.com/report/genetic-toxicology-testing-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model