Human Immunodeficiency Viruses (HIV) Clinical Trials Market Forecast 2026: Where the Industry Is Headed
Uncover key drivers, emerging technologies, and competitive movements shaping the human immunodeficiency viruses (hiv) clinical trials market from 2026–2035 with trusted insights from The Business Research Company
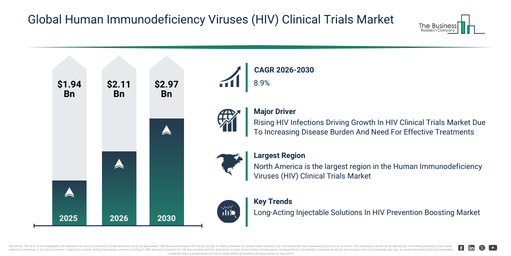
What are the forecasted starting (2026) and ending (2030) market sizes for the Human Immunodeficiency Viruses (HIV) Clinical Trials Market?
The human immunodeficiency viruses (hiv) clinical trials market size has demonstrated significant growth in recent years. It is anticipated to expand from $1.94 billion in 2025 to $2.11 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 9.2%. The historical increase in this market can be ascribed to a high global hiv disease burden, the broadening of antiretroviral drug research, increased public and private funding for hiv studies, the expansion of clinical research infrastructure, and regulatory support for hiv drug development.
The human immunodeficiency viruses (hiv) clinical trials market size is anticipated to demonstrate robust expansion over the forthcoming years. It is projected to achieve a valuation of $2.97 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 8.9%. This growth during the forecast period is attributable to the increasing development of curative and preventive hiv therapies, a rising adoption of adaptive trial designs, an expansion of vaccine-focused hiv trials, the growing application of AI in clinical research, and an increased emphasis on long-term safety and efficacy outcomes. Prominent trends for the forecast period include the increasing adoption of decentralized clinical trial models, a rising utilization of digital patient recruitment tools, a growing focus on long-acting hiv therapies within trials, an expansion of real-world evidence integration, and enhanced use of data analytics in trial management.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28244&type=smp
What Drivers Are Supporting Technological Adoption In The Human Immunodeficiency Viruses (HIV) Clinical Trials Market?
The rising prevalence of human immunodeficiency virus (HIV) infections is anticipated to stimulate the expansion of the human immunodeficiency virus (HIV) clinical trials market in the coming period. Human immunodeficiency virus (HIV) infections describe a condition where the HIV virus targets and weakens the immune system, making individuals more susceptible to illnesses and potentially progressing to AIDS. This increase in HIV infections is largely due to insufficient understanding of transmission, leading to high-risk behaviors and delayed diagnosis, which facilitate the virus’s spread. Human immunodeficiency virus (HIV) clinical trials support patients with HIV infections by evaluating new medications, vaccines, and therapeutic strategies to enhance prevention, more effectively manage the virus, and ultimately reduce disease advancement and transmission. For instance, in August 2024, data from the Minority HIV/AIDS Fund (MHAF), a US-based government agency, revealed that an estimated 39.9 million individuals globally were living with HIV in 2023, comprising 38.6 million adults and 1.4 million children, a figure comparable to previous years. Thus, the growing incidence of human immunodeficiency virus (HIV) infections is propelling the growth of the human immunodeficiency virus (HIV) clinical trials market.
What Segment Types Are Examined In The Human Immunodeficiency Viruses (HIV) Clinical Trials Market?
The human immunodeficiency viruses (hiv) clinical trials market covered in this report is segmented –
1) By Phase: Phase I, Phase II, Phase III, Phase IV, Preclinical
2) By Drug Type: Antiretroviral Therapy (ART), Pre-Exposure Prophylaxis (PrEP), Post-Exposure Prophylaxis (PEP), Long-Acting Injectable Antiretrovirals, Experimental Drugs
3) By Study Design: Interventional Studies, Observational Studies, Expanded Access Studies
4) By Patient Population: Adults, Pediatric Patients, Seniors, Pregnant Women, High-Risk Populations
5) By End User: Pharmaceutical Companies, Research Institutes, Hospitals, Other End-Users
Subsegments:
1) By Phase I: First-In-Human Studies, Safety And Tolerability Studies
2) By Phase II: Dose-Finding Studies, Efficacy Studies
3) By Phase III: Pivotal Trials, Comparative Efficacy Studies
4) By Phase Iv: Post-Marketing Surveillance, Long-Term Safety Studies
5) By Preclinical: In Vitro Studies, Animal Studies
Which Trends Are Expected To Influence The Human Immunodeficiency Viruses (HIV) Clinical Trials Market In The Upcoming Years?
Leading companies in the human immunodeficiency virus (HIV) clinical trials market are concentrating on developing innovative solutions like HIV prevention injections to boost treatment effectiveness and lower transmission rates. An HIV prevention injection is defined as a long-acting injectable medication given periodically to prevent HIV infection in high-risk individuals by preventing the virus from establishing itself in the body. For instance, in August 2025, Gilead Sciences Inc., a US-based pharmaceutical company, received European Union approval for Yeytuo (lenacapavir) for HIV treatment. This twice-yearly injectable HIV prevention treatment demonstrates high efficacy, almost 100% in clinical trials, substantially reducing the risk of sexually acquired HIV-1 infection. Its long-acting dosage improves adherence and convenience compared to daily oral PrEP, thereby filling a significant gap in HIV prevention. Designed for adults and adolescents at increased risk, it aims to expand access and help reduce new HIV infections globally as part of comprehensive prevention strategies.
Who Are The Primary Competitors In The Global Human Immunodeficiency Viruses (HIV) Clinical Trials Market?
Major companies operating in the human immunodeficiency viruses (hiv) clinical trials market are Johnson & Johnson, F. Hoffmann-La Roche Ltd., Pfizer Inc., AbbVie Inc., Sanofi S.A., Thermo Fisher Scientific Inc., Amgen Inc., Gilead Sciences Inc., Merck & Co., IQVIA Holdings Inc., ICON plc, Moderna Inc., ViiV Healthcare Limited, HOOKIPA Pharma Inc., Excision BioTherapeutics Inc., American Gene Technologies International Inc., Bionor Holding AS, Aelix Therapeutics, Immuno Cure BioTech Private Limited, ImmunityBio
Access The Complete Report For Deeper Market Insights:
Which Regions Are Poised For Strategic Growth In The Human Immunodeficiency Viruses (HIV) Clinical Trials Market?
North America was the largest region in the human immunodeficiency viruses (HIV) clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the human immunodeficiency viruses (hiv) clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Human Immunodeficiency Viruses (HIV) Clinical Trials Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=28244&type=smp
Browse Through More Reports Similar to the Global Human Immunodeficiency Viruses (HIV) Clinical Trials Market 2026, By The Business Research Company
Virtual Clinical Trials Market Report 2026
https://www.thebusinessresearchcompany.com/report/virtual-clinical-trials-global-market-report
Hiv Drug Market Report 2026
https://www.thebusinessresearchcompany.com/report/hiv-drug-global-market-report
Antiviral Combination Therapy Market Report 2026
https://www.thebusinessresearchcompany.com/report/antiviral-combination-therapy-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model