Vasomotor Menopausal Symptoms (VMS) Treatment Market Anticipated to Record Steady Gains, Advancing to $26.47 Billion by 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the vasomotor menopausal symptoms (vms) treatment market from 2026–2035 with trusted insights from The Business Research Company
What long-term size projections exist for the Vasomotor Menopausal Symptoms (VMS) Treatment Market covering 2026–2035?
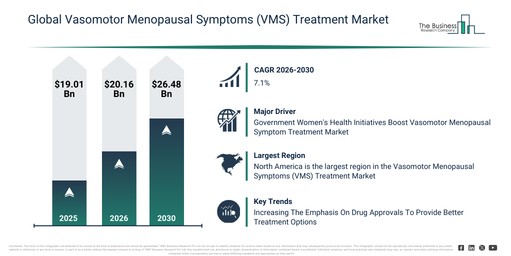
The vasomotor menopausal symptoms (vms) treatment market size has seen robust expansion in recent years. This market is projected to climb from $19.01 billion in 2025 to $20.16 billion in 2026, registering a compound annual growth rate (CAGR) of 6.0%. The market’s historical growth can be attributed to an increasing prevalence of menopausal symptoms, the widespread adoption of hormone replacement therapy, expanding clinical research into neurotransmitter-based treatments, the development of women’s health services, and improved access to specialty clinics.
The vasomotor menopausal symptoms (vms) treatment market is anticipated to show significant expansion over the next few years, with its size expected to reach $26.48 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 7.1%. This growth during the forecast period stems from factors such as increasing demand for non-hormonal treatment alternatives, heightened awareness regarding menopause-related health management, growing investments in women’s health research, the expansion of telemedicine-based treatment delivery, and the development of next-generation VMS therapeutics. Major trends during this period are projected to include the accelerating development of non-hormonal therapies, the increasing adoption of personalized menopause treatment plans, a rising reliance on digital health platforms for symptom management, the expansion of combination drug therapies, and an enhanced focus on long-term safety profiles.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=13051&type=smp
Which Drivers Are Impacting The Competitive Landscape Of The Vasomotor Menopausal Symptoms (VMS) Treatment Market?
Government-led initiatives concerning women’s health during menopause are anticipated to stimulate the expansion of the vasomotor menopausal symptom’s treatment market. These governmental programs for menopausal health include financial support for innovative studies on menopausal transition, illness prevention, and research encompassing pharmacological, clinical, and epidemiological facets. These efforts are integral to wider women’s health initiatives designed to address diverse concerns. For instance, in January 2023, the Government of New South Wales, an Australia-based government agency, reported that it allocated $40.3 million over four years (2022-2023 to 2025-2026) to establish up to 16 new programs for women experiencing severe menopausal symptoms. Thus, the government’s initiatives for women’s health in menopause are actively driving the growth of the vasomotor menopausal symptom’s treatment market.
What Leading Segments Are Studied In The Vasomotor Menopausal Symptoms (VMS) Treatment Market?
The vasomotor menopausal symptoms (vms) treatment market covered in this report is segmented –
1) By Drug Class: Antidepressants, Fluoxetine, Paroxetine, Sertraline, Hormone Therapy, Anticonvulsant, Other Drug Classes
2) By Distribution Channel: Hospital Pharmacy, Retail Pharmacy, Online Pharmacy, Other Distribution Channels
3) By End-User: Hospitals, Specialty Clinics, Homecare, Other End-Users
Subsegments:
1) By Antidepressants: SSRIs (Selective Serotonin Reuptake Inhibitors), SNRIs (Serotonin-Norepinephrine Reuptake Inhibitors), TCAs (Tricyclic Antidepressants), MAOIs (Monoamine Oxidase Inhibitors), Atypical Antidepressants
2) By Fluoxetine: Depression, Obsessive-Compulsive Disorder (OCD), Panic Disorder, Generalized Anxiety Disorder (GAD), Post-Traumatic Stress Disorder (PTSD), Social Anxiety Disorder, Premenstrual Dysphoric Disorder (PMDD)
3) By Paroxetine: Depression, Obsessive-Compulsive Disorder (OCD), Panic Disorder, Generalized Anxiety Disorder (GAD), Post-Traumatic Stress Disorder (PTSD), Social Anxiety Disorder, Premenstrual Dysphoric Disorder (PMDD)
4) By Sertraline: Depression, Obsessive-Compulsive Disorder (OCD), Panic Disorder, Post-Traumatic Stress Disorder (PTSD), Social Anxiety Disorder, Premenstrual Dysphoric Disorder (PMDD)
5) By Hormone Therapy: Estrogen Replacement Therapy (ERT), Progestin Therapy, Combined Hormonal Therapy (CHT), Testosterone Replacement Therapy (TRT)
6) By Anticonvulsant: Hydantoins, Barbiturates, Benzodiazepines, Valproates, Succinimides, Carboxylic Acid Derivatives
7) By Other Drug Classes: Antibiotics, Analgesics, Antidiabetics, Antihypertensives, Anticoagulants, Immunosuppressants, Antipsychotics, Antihistamines
What Trends Are Driving The Growth Trajectory Of The Vasomotor Menopausal Symptoms (VMS) Treatment Market?
Leading companies in the vasomotor menopausal symptoms treatment market are actively developing products, such as Veozah, and securing regulatory approvals to introduce new therapeutic choices and address patients’ unmet medical needs. The authorization of novel medications for menopause offers superior efficacy and safety when compared to existing treatments. For instance, in May 2023, Astellas Pharma Inc, a Japan-based pharmaceutical company, obtained approval for Veozah (fezolinetant) 45 mg once daily from the Food and Drug Administration (FDA), a US-based federal agency, for the management of menopausal vasomotor symptoms (VMS), which vary from mild to severe. This represents the first nonhormonal neurokinin 3 (NK3) receptor antagonist to receive authorization for treating menopausal-related VMS. It functions as a hormone-free solution by blocking a specific pathway in the brain that helps control these symptoms. Veozah provides an alternative treatment option for women experiencing these symptoms, particularly those who are unable to use or prefer to avoid hormone therapy. The approval of Veozah underscores its significance as a new drug for managing menopausal symptoms.
Which Major Firms Are Strengthening Their Position In The Vasomotor Menopausal Symptoms (VMS) Treatment Market?
Major companies operating in the vasomotor menopausal symptoms (vms) treatment market are Pfizer Inc., Abbvie Inc., Novartis AG, Teva Pharmaceutical Industries Ltd., Mylan N.V., Sun Pharmaceutical Industries Ltd., Aurobindo Pharma Limited, Kyowa Kirin Co. Ltd., Dr. Reddy’s Laboratories Ltd., Alembic Pharmaceuticals Limited Inc., Apotex Inc., Hikma Pharmaceutical plc, Endo Pharmaceuticals plc, Amneal Pharmaceuticals LLC, Mundipharma International Ltd., Lupin Limited, Glenmark Pharmaceuticals Ltd., Hisamitsu Pharmaceutical Co. Inc., Emcure Pharmaceuticals Limited, Wockhardt Pvt. Ltd., QuatRx Pharmaceuticals Company, Bionovo Inc., Mithra Pharmaceuticals SA, EndoCeutics Inc., Zydus Group, Edgemont Pharmaceuticals LLC, MenoGeniX Inc., Agile Therapeutics Inc
Access The Complete Report For Deeper Market Insights:
Which Regions Are Poised For Strategic Growth In The Vasomotor Menopausal Symptoms (VMS) Treatment Market?
North America was the largest region in the vasomotor menopausal symptoms (VMS) treatment market in 2025. The regions covered in the vasomotor menopausal symptoms (vms) treatment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Vasomotor Menopausal Symptoms (VMS) Treatment Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=13051&type=smp
Browse Through More Reports Similar to the Global Vasomotor Menopausal Symptoms (VMS) Treatment Market 2026, By The Business Research Company
Vulvodynia Treatment Market Report 2026
https://www.thebusinessresearchcompany.com/report/vulvodynia-treatment-global-market-report
Menopausal Hot Flashes Market Report 2026
https://www.thebusinessresearchcompany.com/report/menopausal-hot-flashes-global-market-report
Menopause Market Report 2026
https://www.thebusinessresearchcompany.com/report/menopause-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model