Messenger Ribo Nucleic Acid (mRNA) Vaccine Contract Development And Manufacturing Organization (CDMO) Market Forecast 2026–2035: How to Leverage Market Growth
Uncover key drivers, emerging technologies, and competitive movements shaping the messenger ribo nucleic acid (mrna) vaccine contract development and manufacturing organization (cdmo) market from 2026–2035 with trusted insights from The Business Research Company
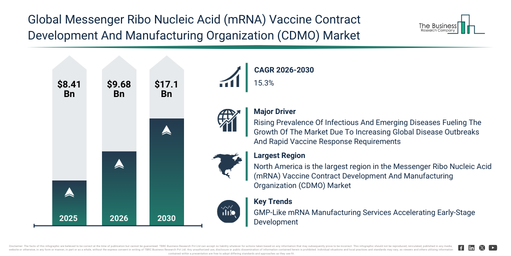
How much larger will the Messenger Ribo Nucleic Acid (mRNA) Vaccine Contract Development And Manufacturing Organization (CDMO) Market be in 2030 compared with 2026?
The messenger ribo nucleic acid (mrna) vaccine contract development and manufacturing organization (cdmo) market has experienced rapid expansion in recent years. This market is projected to grow from $8.41 billion in 2025 to $9.68 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 15.1%. The historical increase in this sector can be attributed to covid 19 vaccine programs, the proliferation of biotech startups, limited in house manufacturing capacity, an uplift in contract manufacturing, and swift mrna platform validation.
The messenger ribo nucleic acid (mrna) vaccine contract development and manufacturing organization (cdmo) market is anticipated to experience significant expansion in the coming years. This market is projected to reach $17.1 billion by 2030, growing at a compound annual growth rate (CAGR) of 15.3%. The drivers for this growth during the forecast period include the increasing adoption of mrna cancer vaccines, the emergence of personalized vaccines, a rise in CDMO collaborations, the necessity for rapid manufacturing capabilities, and an expanding portfolio of mrna therapies. Noteworthy trends in the forecast period encompass lipid nanoparticle formulation services, swift mrna process scale-up, integrated fill-finish outsourcing, flexible modular vaccine manufacturing suites, and comprehensive end-to-end mrna development offerings.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=33214&type=smp
What Drivers Are Guiding Growth Patterns In The Messenger Ribo Nucleic Acid (mRNA) Vaccine Contract Development And Manufacturing Organization (CDMO) Market?
The increasing occurrence of infectious and newly appearing diseases is anticipated to drive the expansion of the mRNA vaccine CDMO market in the future. These diseases are defined as illnesses caused by disease-causing microorganisms that either spread quickly or emerge recently, influenced by factors like environmental shifts, urbanization, and enhanced global connections. Increased global travel, higher population densities, and interwoven supply chains have quickened the spread of pathogens, resulting in more common, widespread, and challenging-to-control disease outbreaks. mRNA vaccine CDMOs are crucial in tackling these issues, facilitating the swift development, mass production, and adaptable manufacturing of vaccines essential for combating evolving infectious dangers. Given that mRNA technologies demand specific manufacturing facilities and specialized technical knowledge, vaccine developers depend on contract development and manufacturing organizations to speed up processes and guarantee manufacturing readiness during disease outbreaks. For example, in May 2024, the World Health Organization (WHO), a global health agency based in Switzerland, reported over 7.6 million dengue cases worldwide as of 30 April 2024. This included 3.4 million confirmed cases, more than 16,000 severe cases, and over 3,000 fatalities. This rise was notably significant in the Region of the Americas, with cases surpassing seven million by April 2024, exceeding the 2023 annual total of 4.6 million cases. Consequently, the increased occurrence of infectious and emerging diseases is fueling the expansion of the mRNA vaccine CDMO market.
Which Segments Are Driving Activity In The Messenger Ribo Nucleic Acid (mRNA) Vaccine Contract Development And Manufacturing Organization (CDMO) Market?
The messenger ribo nucleic acid (mrna) vaccine contract development and manufacturing organization (cdmo) market covered in this report is segmented –
1) By Service Type: Process Development; Manufacturing; Analytical And Quality Control; Fill Finish; Packaging; Other Service Types
2) By Vaccine Type: COVID-19 Vaccines; Cancer Vaccines; Infectious Disease Vaccines; Other Vaccine Types
3) By Scale of Operation: Preclinical; Clinical; Commercial
4) By End-User: Pharmaceutical Companies; Biotechnology Companies; Research Institutes; Other End-Users
Subsegments:
1) By Process Development: Formulation Optimization; Cell Line Development; Upstream Process Development; Process Scale-Up
2) By Manufacturing: Lipid Nanoparticle Production; Enzymatic Capping; Purification Process
3) By Analytical And Quality Control: Potency Testing; Purity Analysis; Stability Testing; Identity Testing; Microbial Testing
4) By Fill Finish: Aseptic Filling; Vial Filling; Syringe Filling; Lyophilization
5) By Packaging: Primary Packaging; Secondary Packaging; Labeling Operations; Cold Chain Packaging; Unit Dose Packaging
6) By Other Service Types: Regulatory Support Services; Technology Transfer Services; Contract Development Services; Consulting Services; Supply Chain Management
What Trends Are Projected To Support The Growth Of The Messenger Ribo Nucleic Acid (mRNA) Vaccine Contract Development And Manufacturing Organization (CDMO) Market?
Companies operating in the mRNA vaccine CDMO market are increasingly concentrating on advanced innovations, such as phase-appropriate GMP mRNA contract development and manufacturing services, to accelerate preclinical and IND-enabling development while upholding regulatory-aligned quality standards and shorter timelines. Phase-appropriate GMP-like mRNA manufacturing refers to a production method designed to supply high-quality, regulatory-conscious mRNA material suitable for preclinical and early development stages, without the expenses and operational complexity associated with full GMP manufacturing. For instance, in May 2025, GenScript Biotech Corporation, a China-based global biotechnology reagents and services provider, launched a GMP-like mRNA manufacturing service aimed at supporting preclinical and IND-enabling development. This service delivers high-quality, regulatory-aware mRNA with reduced lead times and lower costs compared to traditional full GMP manufacturing, reflecting the growing demand for flexible, phase-appropriate CDMO solutions in the mRNA vaccine market.
Which Major Firms Are Strengthening Their Position In The Messenger Ribo Nucleic Acid (mRNA) Vaccine Contract Development And Manufacturing Organization (CDMO) Market?
Major companies operating in the messenger ribo nucleic acid (mrna) vaccine contract development and manufacturing organization (cdmo) market are Sanofi S.A., Thermo Fisher Scientific Inc., FUJIFILM Diosynth Biotechnologies U.S.A. Inc., Danaher Corporation, Boehringer Ingelheim BioXcellence GmbH & Co. KG, AGC Biologics A/S, Eurofins Scientific SE, Lonza Group AG, BioNTech SE, Samsung Biologics Co. Ltd., WuXi Biologics (Cayman) Inc., Siegfried Holding AG, Recipharm AB, Vetter Pharma International GmbH, CordenPharma GmbH & Co. KG, Ajinomoto Bio-Pharma Services Inc., Novasep Holding S.A.S., Rentschler Biopharma SE, IDT Biologika GmbH, Wacker Biotech GmbH
Access The Complete Report For Deeper Market Insights:
Which Region Shows The Strongest Potential For Future Expansion In The Messenger Ribo Nucleic Acid (mRNA) Vaccine Contract Development And Manufacturing Organization (CDMO) Market?
North America was the largest region in the mRNA vaccine contract development and manufacturing organization (CDMO) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the messenger ribo nucleic acid (mrna) vaccine contract development and manufacturing organization (cdmo) market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Messenger Ribo Nucleic Acid (mRNA) Vaccine Contract Development And Manufacturing Organization (CDMO) Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=33214&type=smp
Browse Through More Reports Similar to the Global Messenger Ribo Nucleic Acid (mRNA) Vaccine Contract Development And Manufacturing Organization (CDMO) Market 2026, By The Business Research Company
Vaccine Contract Manufacturing Market Report 2026
Mrna Cancer Vaccines And Therapeutics Market Report 2026
Therapeutic Bcg Vaccines Market Report 2026
https://www.thebusinessresearchcompany.com/report/therapeutic-bcg-vaccines-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model