Global Pharmaceutical Quality Control Release Testing Service Market Performance Report: Global Trends 2026–2030
Uncover key drivers, emerging technologies, and competitive movements shaping the pharmaceutical quality control release testing service market from 2026–2035 with trusted insights from The Business Research Company
What long-term size projections exist for the Pharmaceutical Quality Control Release Testing Service Market covering 2026–2035?
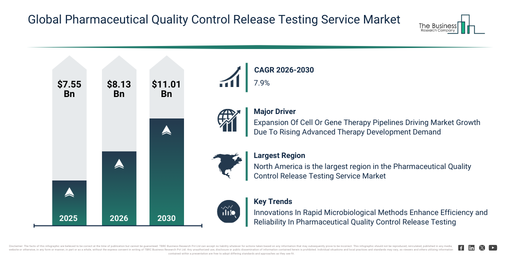
The pharmaceutical quality control release testing service market size has demonstrated significant growth in recent years. It is projected to increase from $7.55 billion in 2025 to $8.13 billion in 2026, at a compound annual growth rate (CAGR) of 7.6%. This expansion in the past can be primarily linked to more rigorous GMP regulations, a surge in global drug production, the proliferation of contract testing laboratories, a greater need for biologics testing, and the ongoing growth of generic drug manufacturing.
The pharmaceutical quality control release testing service market size is projected to experience substantial expansion over the upcoming period. This market is forecast to reach $11.01 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 7.9%. This anticipated growth during the forecast timeframe is driven by factors such as the expanding pipelines of complex biologics, an increase in regulatory audit occurrences, the broadening adoption of outsourced quality control models, the need for quicker batch release cycles, and a rise in multi-region drug approvals. Key trends emerging in this forecast period encompass automated batch release testing, digital quality systems for laboratories, analytical validation supported by AI, interconnected stability testing laboratories, and intelligent quality data platforms.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=33260&type=smp
Which Drivers Are Impacting The Competitive Landscape Of The Pharmaceutical Quality Control Release Testing Service Market?
The growing number of cell or gene therapy pipelines is anticipated to fuel the expansion of the pharmaceutical quality control release testing service market moving forward. These pipelines represent cellular and genetic therapeutic products currently undergoing development and regulatory assessment for commercial release. This expansion is largely fueled by heightened investment in biotechnology research and development, as pharmaceutical and biotech companies commit significant resources to pioneering cellular and genetic treatments for conditions previously lacking effective therapies. The increasing cell and gene therapy pipeline directly boosts the need for specialized quality control release testing services, which are crucial to ensure that these intricate biological products adhere to rigorous safety and efficacy benchmarks prior to their market introduction. For instance, in January 2025, the American Society of Gene and Cell Therapy, a US-based scientific association, reported that the global gene therapy pipeline reached 2,117 programs in the fourth quarter of 2024, up from 2,041 programs in the third quarter of 2024. Consequently, the expansion of cell or gene therapy pipelines is a key factor propelling the growth of the pharmaceutical quality control release testing service market.
What Segments Are Identified Within The Structure Of The Pharmaceutical Quality Control Release Testing Service Market?
The pharmaceutical quality control release testing service market covered in this report is segmented –
1) By Product Type: Dissolution Testing; Anti-Corrosion Testing; ICH Stability Storage and Testing; Other Product Types
2) By Service Type: Analytical Testing Services; Microbiological Testing Services; Stability Testing Services; Sterility Testing Services
3) By Technology: Gas Chromatography (GC); Mass Spectrometry; Spectroscopy Methods
4) By End Users: Pharmaceutical Manufacturers; Biotechnology Companies; Contract Development and Manufacturing Organizations (CDMOs); Contract Manufacturing Organizations (CMOs); Research and Academic Institutions
Subsegments:
1) By Dissolution Testing: Immediate Release Testing; Extended Release Testing; Delayed Release Testing; Sink Condition Evaluation
2) By Anti-Corrosion Testing: Packaging Material Corrosion Testing; Container Compatibility Assessment; Environmental Stress Corrosion Testing; Protective Coating Evaluation
3) By ICH Stability Storage and Testing: Long Term Stability Studies; Accelerated Stability Studies; Intermediate Stability Studies; Photostability Testing
4) By Other Product Types: Impurity Profiling; Residual Solvent Testing; Elemental Impurity Analysis; Container Closure Integrity Testing
What Upcoming Trends Are Likely To Define The Future Path Of The Pharmaceutical Quality Control Release Testing Service Market?
Leading companies operating within the pharmaceutical quality control release testing services market are prioritizing the development of advanced solutions, including microbiological-based quality control testing. This focus aims to ensure product sterility, maintain regulatory compliance, and facilitate the timely release of batches. Microbiological-based quality control testing utilizes microbiological techniques to effectively monitor and measure the presence of microbes in both pharmaceutical products and manufacturing settings. This approach is crucial for confirming sterility, safeguarding product safety, and meeting regulatory requirements before products are released. As an example, in March 2025, Nelson Laboratories, a US-based microbiological testing company, introduced its Rapid Sterility Testing. This microbiology-based quality control testing offering was designed to modernize sterility assurance processes and support the faster batch release of sterile pharmaceutical products. The solution effectively shortens traditional fourteen-day incubation timelines to approximately six days and incorporates automated, quantitative data analytics to enhance result precision, reduce manual intervention, and improve overall testing reliability.
Which Leading Companies Dominate The Pharmaceutical Quality Control Release Testing Service Market Share?
Major companies operating in the pharmaceutical quality control release testing service market are Labcorp Holdings Inc., SGS S.A., Eurofins Scientific SE, Selvita S.A., WuXi AppTec Co. Ltd., Intertek Group plc, Charles River Laboratories International Inc., Piramal Pharma Limited, Siegfried Holding AG, Almac Group Limited, NSF International, ClinChoice Inc., BioAgilytix Inc., Microbac Laboratories Inc., ARL Bio Pharma Inc., Quality Chemical Laboratories LLC, Neopharm Labs Inc., Butterworth Laboratories Limited, Cambridge Polymer Group Inc., Honeyman Group Limited, and Element Materials Technology Group Limited.
Access The Complete Report For Deeper Market Insights:
How Does The Pharmaceutical Quality Control Release Testing Service Market Perform Across Major Global Regions?
North America was the largest region in the pharmaceutical quality control release testing service market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the pharmaceutical quality control release testing service market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Pharmaceutical Quality Control Release Testing Service Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=33260&type=smp
Browse Through More Reports Similar to the Global Pharmaceutical Quality Control Release Testing Service Market 2026, By The Business Research Company
Pharmaceutical Packaging Testing Equipment Market Report 2026
Pharma Admet Testing Market Report 2026
https://www.thebusinessresearchcompany.com/report/pharma-admet-testing-global-market-report
Pharmaceutical Label Market Report 2026
https://www.thebusinessresearchcompany.com/report/pharmaceutical-label-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model