Biologics Sterile Fill-Finish Services Market Growth Projection: Expected to Surpass $8.35 Billion by 2030 at 10.7% CAGR
Uncover key drivers, emerging technologies, and competitive movements shaping the biologics sterile fill-finish services market from 2026–2035 with trusted insights from The Business Research Company
What total market size is anticipated for the Biologics Sterile Fill-Finish Services Market in 2030?
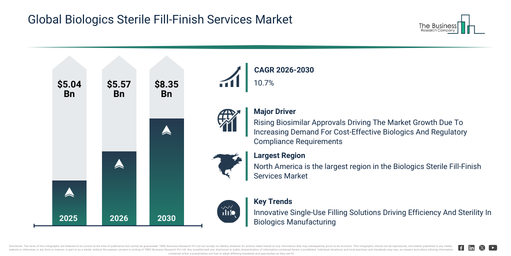
The biologics sterile fill-finish services market has seen rapid growth in recent years. Its valuation is projected to rise from $5.04 billion in 2025 to $5.57 billion in 2026, at a compound annual growth rate (CAGR) of 10.4%. This historical expansion can be attributed to the broadening of biologics clinical trial pipelines, the increasing commercialization of injectable biologics, the expansion of vaccine production programs, a rise in the outsourcing of fill-finish services, and the tightening of regulatory requirements for sterility.
The biologics sterile fill-finish services market size is poised for substantial growth over the next few years, projected to reach $8.35 billion by 2030, with a compound annual growth rate (CAGR) of 10.7%. This expansion in the forecast period is driven by factors such as the increasing demand for commercial-scale biologics filling, the rising adoption of advanced aseptic technologies, the expansion of high-potency biologic products, a growing focus on flexible and modular fill-finish facilities, and increasing serialization and traceability requirements. Throughout this period, major trends will include the increasing adoption of automated aseptic filling lines, a rising demand for high-capacity lyophilization services, the expansion of prefilled syringe filling capabilities, a growing emphasis on contamination control, and the enhanced use of isolator and RABS technologies.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=33041&type=smp
What Drivers Are Influencing Production Trends In The Biologics Sterile Fill-Finish Services Market?
The biologics sterile fill-finish services market is projected to expand propelled by the increase in biosimilar approvals. Biosimilar approvals signify official regulatory authorizations that allow a biosimilar medicine to be marketed and used as equivalent to its reference biologic. The escalation in biosimilar approvals stems from a heightened demand for economical alternatives to costly biologics, thereby improving patient access and reducing overall healthcare expenditures. Biologics sterile fill-finish services aid biosimilar approvals by ensuring that the final drug product is produced safely, is sterile, and is consistent, thereby fulfilling the rigorous regulatory quality standards required for approval and use in clinical settings. For instance, in September 2025, data from the MBIP, an Australia-based government department, indicated that Australian biosimilars numbered 65 in 2024, with a notable 22 additional approvals occurring in that year alone. Furthermore, in 2024, according to Cardinal Health Inc., a US-based healthcare company, a total of 46 biosimilars had secured FDA approval by 2023. Thus, the rising number of biosimilar approvals is a significant driver for the growth of the biologics sterile fill-finish services market.
Which Segment Groups Are Influencing The Biologics Sterile Fill-Finish Services Market?
The biologics sterile fill-finish services market covered in this report is segmented –
1) By Service Type: Liquid Filling; Lyophilization; Aseptic Filling; Labeling And Packaging
2) By Container Type: Glass Containers; Plastic Containers; Pre Filled Syringes; Bulk Bags
3) By Technology: Automated Filling Systems; Manual Filling Systems; Isolator Technology; Restricted Access Barrier Systems; Advanced Aseptic Processing Technologies
4) By End User: Pharmaceutical Companies; Biotechnology Firms; Contract Development And Manufacturing Organizations; Academic And Research Institutions; Government Agencies
Subsegments:
1) By Liquid Filling: Vial Liquid Filling; Syringe Liquid Filling; Cartridge Liquid Filling; Bottle Liquid Filling; Ampoule Liquid Filling
2) By Lyophilization: Single Chamber Lyophilization; Dual Chamber Lyophilization; Controlled Rate Lyophilization; High Capacity Lyophilization; Continuous Lyophilization
3) By Aseptic Filling: Vial Aseptic Filling; Syringe Aseptic Filling; Cartridge Aseptic Filling; Ampoule Aseptic Filling; Bag Aseptic Filling
4) By Labeling And Packaging: Primary Packaging; Secondary Packaging; Tertiary Packaging; Serialized Packaging; Tamper Evident Packaging
Which Trends Are Influencing The Performance And Direction Of The Biologics Sterile Fill-Finish Services Market?
Leading companies active in the biologics sterile fill-finish services market are concentrating on developing advanced solutions, such as single-use filling systems, to enhance production efficiency, mitigate contamination risks, and support flexible manufacturing of biologics. A single-use filling system denotes a pre-sterilized, disposable setup employed in biologics manufacturing that allows for the direct filling of drug products without requiring repeated cleaning, thereby minimizing cross-contamination and downtime. For example, in October 2025, Shenzhen China Resources Gosun Pharmaceutical Co. Ltd., a China-based pharmaceutical enterprise, launched a biologics aseptic fill-finish line to augment its capacity for sterile biologic drug production. This line directly facilitates sterile fill-finish services for biologics, utilizing over 30 years of expertise with automated SKAN isolators, single-use systems, and 100% in-line checks to assure zero contamination, minimal waste, and GMP compliance. Its advantages include decreased human intervention, precise filling without shear stress, quicker scalability, and high yields for sensitive products like monoclonal antibodies. The fundamental aim is to bridge upstream biologics production to safe, sterile, patient-ready injectables amidst increasing CDMO demand.
Who Are The Major Companies Operating In The Biologics Sterile Fill-Finish Services Market?
Major companies operating in the biologics sterile fill-finish services market are Thermo Fisher Scientific Inc., Baxter International Inc., Lonza Group AG, Samsung Biologics, WuXi Biologics, Recipharm AB, Vetter Pharma‑Fertigung GmbH & Co. KG, Emergent BioSolutions Inc., Aenova Holding GmbH, Ajinomoto Bio‑Pharma Services, Novasep Group, Grand River Aseptic Manufacturing, Akron Biotech Inc., Seikagaku Corporation, Jubilant HollisterStier LLC, Boehringer Ingelheim BioXcellence, Symbiosis Pharmaceutical Services Ltd., HALIX B.V., Afton Scientific Inc., Jiangsu YaoHAI Bio‑Pharmaceutical Co. Ltd., Cognate BioServices, TAIYO Pharma Tech Co. Ltd.
Access The Complete Report For Deeper Market Insights:
Which Region Is Anticipated To See The Fastest Growth In The Biologics Sterile Fill-Finish Services Market?
North America was the largest region in the biologics sterile fill-finish services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the biologics sterile fill-finish services market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Biologics Sterile Fill-Finish Services Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=33041&type=smp
Browse Through More Reports Similar to the Global Biologics Sterile Fill-Finish Services Market 2026, By The Business Research Company
Sterile Medical Packaging Market Report 2026
https://www.thebusinessresearchcompany.com/report/sterile-medical-packaging-global-market-report
Depyrogenated Sterile Empty Vials Market Report 2026
Generic Oncology Sterile Injectable Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model
[KClientError] [REQ_ERR: OPERATION_TIMEDOUT] [KTrafficClient] Something is wrong.