Early Phase Clinical Trial Outsourcing Market 2026–2030: Emerging Trends and Forecast Opportunities
Uncover key drivers, emerging technologies, and competitive movements shaping the early phase clinical trial outsourcing market from 2026–2035 with trusted insights from The Business Research Company
Across 2026–2030, what is the expected market valuation path of the Early Phase Clinical Trial Outsourcing Market?
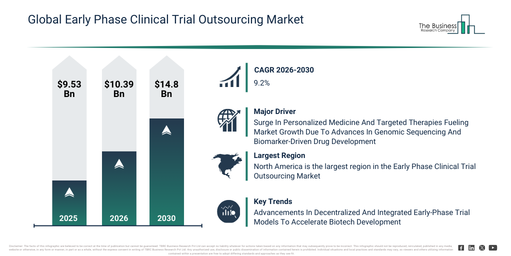
The early phase clinical trial outsourcing market has experienced robust growth in recent years. It is anticipated to increase from $9.53 billion in 2025 to $10.39 billion in 2026, at a compound annual growth rate (CAGR) of 9.0%. The expansion observed historically can be ascribed to the increasing complexity of drug development pipelines, rising cost pressures on pharmaceutical sponsors, the proliferation of biotech startup activity, an increasing need for regulatory expertise, and a greater reliance on specialized clinical research organizations.
The early phase clinical trial outsourcing market size is anticipated to undergo significant expansion in the coming years. It is projected to climb to $14.8 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 9.2%. This projected growth can be attributed to the increasing adoption of decentralized trial elements, a rising focus on precision medicine studies, the expansion of rare disease clinical research, the growing utilization of AI-enabled trial analytics, and increasing global harmonization of early-phase regulations. Prominent trends within the forecast period include the increasing outsourcing of phase I clinical studies, a rising demand for integrated early-phase contract research organization (CRO) services, the growing application of data-driven trial design approaches, the expansion of adaptive and first-in-human trials, and an enhanced focus on accelerated study timelines.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=33097&type=smp
Which Drivers Are Supporting The Rise Of The Early Phase Clinical Trial Outsourcing Market?
The early-phase clinical trial outsourcing market is anticipated to expand moving forward, driven by the escalating attention to personalized medicine. Personalized medicine denotes the heightened focus within healthcare and life sciences on developing and delivering treatments precisely tailored to individual patients or clearly defined patient subgroups based on their genetic and molecular characteristics. This increasing emphasis on personalized medicine primarily stems from advancements in genomic sequencing and molecular diagnostics, which enable the accurate identification of genetic and biomarker profiles specific to diseases. Early-phase clinical trial outsourcing supports personalized medicine and targeted therapies by offering specialized expertise, flexible trial designs, and biomarker-driven study capabilities, facilitating precise patient stratification and rapid proof-of-concept validation. For instance, in February 2024, according to the Personalized Medicine Coalition (PMC), a US-based nonprofit organization, the count of personalized therapies for rare diseases more than doubled, climbing from 6 in 2022 to 16 in 2023. Hence, the increasing focus on personalized medicine is a key factor propelling the growth of the early-phase clinical trial outsourcing market.
What Segments Are Identified Within The Structure Of The Early Phase Clinical Trial Outsourcing Market?
The early phase clinical trial outsourcing market covered in this report is segmented –
1) By Type: Regulatory Services; Clinical Data Management (CDM); Medical Writing; Site Management; Pharmacovigilance (PV); Risk-Based Monitoring; Bio Statistical Services; Protocol Development; Other Types
2) By Therapeutic Area: Oncology; Central Nervous System (CNS) And Neurology; Cardiovascular And Metabolic Diseases; Infectious Diseases; Immunology And Inflammatory Diseases; Rare Diseases And Genetic Disorders
3) By Trial Phase: Phase 0; Phase 1; Phase 2
4) By Applications: Pharmaceutical Companies; Biopharmaceutical Companies; Drug Discovery Companies; Medical Devices Companies; Other Applications
Subsegments:
1) By Regulatory Services: Regulatory Strategy Planning; Regulatory Submission Preparation; Health Authority Liaison; Ethics Committee Submission Management; Regulatory Compliance Auditing; Trial Registration Support
2) By Clinical Data Management (CDM): Case Report Form Design; Data Entry And Validation; Database Development And Maintenance; Data Cleaning And Query Management; Clinical Data Integration; Data Lock And Archival
3) By Medical Writing: Clinical Study Protocol Writing; Investigator Brochure Development; Clinical Study Report Writing; Informed Consent Document Preparation; Regulatory Dossier Writing; Scientific Publication Support
4) By Site Management: Site Feasibility Assessment; Site Selection And Initiation; Investigator Training And Support; Site Performance Management; Site Close Out Services; Vendor Coordination At Site Level
5) By Pharmacovigilance (PV): Safety Data Collection; Adverse Event Processing; Serious Adverse Event Reporting; Safety Database Management; Signal Detection And Evaluation; Periodic Safety Report Preparation
6) By Risk-Based Monitoring: Risk Assessment And Categorization; Centralized Monitoring Activities; Key Risk Indicator Development; Remote Monitoring Services; On Site Monitoring Optimization; Monitoring Plan Design
7) By Bio Statistical Services: Statistical Analysis Plan Development; Sample Size Determination; Randomization Design; Interim Analysis; Final Statistical Analysis; Data Interpretation And Reporting
8) By Protocol Development: Study Design Consulting; Endpoint Definition; Eligibility Criteria Development; Schedule Of Assessments Design; Protocol Amendment Management; Feasibility Driven Protocol Optimization
9) By Other Types: Clinical Trial Supply Management; Patient Recruitment Support; Quality Assurance Services; Trial Master File Management; Vendor And Project Management
How Are Emerging Trends Affecting The Progression Of The Early Phase Clinical Trial Outsourcing Market?
Companies operating within the early-phase clinical trial outsourcing market are emphasizing technological innovations across decentralized and hybrid early-phase clinical trial models, such as integrated early development trial ecosystems. This focus aims to satisfy the growing demand among biotechnology sponsors for more rapid execution, elevated scientific rigor, and reduced risk in early-stage clinical development. Integrated early development trial ecosystems represent specialized clinical research environments that combine dedicated infrastructure, experienced early-phase investigators, real-time safety surveillance, and streamlined regulatory and operational processes, leading to enhanced efficiency and consistency in early-phase trials. As an illustration, in October 2025, Avance Clinical, an Australia-based contract research organization (CRO), announced the opening of a Dedicated Center of Excellence. This center is intended to accelerate early-phase biotech trials, boosting operational efficiency, scientific integrity, and the speed-to-market for emerging biotechnology companies. The Dedicated Center of Excellence provides a centralized early-phase trial setting, integrating specialized clinical units, expert early-phase investigators, and coordinated operational oversight to expedite study start-up and execution.
Which Leading Companies Dominate The Early Phase Clinical Trial Outsourcing Market Share?
Major companies operating in the early phase clinical trial outsourcing market are IQVIA Holdings Inc., ICON plc, Syneos Health Inc., Parexel International Corporation, Medpace Holdings Inc., Pharmaron Beijing Co. Ltd., Premier Research Group Limited, ProPharma Group Holdings Inc., Precision for Medicine Group LLC, Worldwide Clinical Trials LLC, Catalyst Clinical Research LLC, Avance Clinical Pty Ltd, Quanticate International Ltd, Clinilabs Drug Development Corporation, Optimapharm d.o.o., RICe – Richmond Pharmacology Ltd, Novotech Health Holdings Pty Ltd, CMIC Holdings Co. Ltd., Medpace Reference Laboratories Inc., iNGENu CRO Pty Ltd
Access The Complete Report For Deeper Market Insights:
How Does The Early Phase Clinical Trial Outsourcing Market Perform Across Major Global Regions?
North America was the largest region in the early phase clinical trial outsourcing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the early phase clinical trial outsourcing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Early Phase Clinical Trial Outsourcing Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=33097&type=smp
Browse Through More Reports Similar to the Global Early Phase Clinical Trial Outsourcing Market 2026, By The Business Research Company
Biomarker Clinical Phase Outsourcing Services Market Report 2026
Advanced Therapeutics Pharmaceutical Outsourcing Market Report 2026
Clinical Trial Supply And Logistics Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model