Hemophilia B Gene Therapy Market Report 2026: Strategic Insights and Revenue Outlook
Uncover key drivers, emerging technologies, and competitive movements shaping the hemophilia b gene therapy market from 2026–2035 with trusted insights from The Business Research Company
What size range is anticipated for the Hemophilia B Gene Therapy Market from 2026 to 2030?
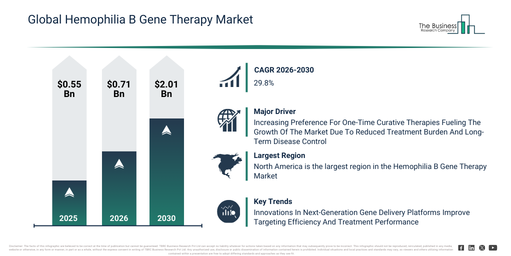
The hemophilia b gene therapy market size has witnessed substantial growth in recent years. It is anticipated to expand from $0.55 billion in 2025 to $0.71 billion in 2026, reflecting a compound annual growth rate (CAGR) of 29.6%. This historical expansion can be attributed to several factors including advancements in gene delivery technologies, a greater comprehension of hemophilia b genetics, the proliferation of clinical research programs, the increasing challenge posed by lifelong replacement therapy, and the availability of specialized treatment centers.
The hemophilia b gene therapy market size is projected to experience substantial growth in the upcoming years. This market is forecast to reach $2.01 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 29.8%. This anticipated growth during the forecast period is driven by several factors, including the increasing number of gene therapy approvals, escalating investments in treatments for rare diseases, a rising need for lasting therapeutic results, the expansion of sophisticated manufacturing capacities, and the growth of patient access initiatives. Key trends anticipated during this period encompass advancements in single-dose curative therapies, a heightened emphasis on sustained Factor IX expression, the continuous refinement of AAV vector designs, the broadening of personalized gene therapy strategies, and improved regulatory harmonization for advanced therapeutic interventions.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=33162&type=smp
What Drivers Are Affecting Demand In The Hemophilia B Gene Therapy Market?
The hemophilia B gene therapy market is projected to expand due to the increasing number of gene therapy clinical trials. These research studies evaluate the safety, effectiveness, and long-term potential of gene-based treatments in humans. The growth in such trials is fueled by greater investment from biopharmaceutical firms, developments in viral vector technology, supportive regulations, and the necessity to address rare genetic conditions with significant unmet medical needs, including hemophilia B. Hemophilia B gene therapy benefits from this expanding landscape by utilizing existing clinical development pathways, regulatory expertise, and manufacturing capabilities linked with in vivo gene delivery methods. For example, in January 2026, the Bioindustry Association (BIA), a UK national trade association, reported 193 ongoing advanced therapy clinical trials in the UK, similar to 2024. Over half were in early stages, approximately 80% were commercially sponsored, and more than 80% concentrated on gene therapies, split equally between ex vivo and in vivo approaches. Consequently, the increasing number of cell and gene therapy clinical trials is stimulating the growth of the hemophilia B gene therapy market. The market’s growth is being driven by the increasing integration of personalized medicine, which facilitates customized treatments, patient categorization, and enhanced therapeutic results. The hemophilia B gene therapy market is anticipated to grow with the increasing adoption of personalized medicine. This medical strategy customizes disease prevention, diagnosis, and treatment based on an individual’s genetic, molecular, or clinical features to improve treatment outcomes and minimize adverse reactions. The greater use of personalized medicine is propelled by advances in genomics, better biomarker identification, and increasing regulatory backing for targeted therapies that move away from universal treatment models. Hemophilia B gene therapy aligns well with personalized medicine principles by addressing the genetic root cause of the condition and providing sustained therapeutic benefits through individualized gene-based interventions. These therapies depend on patient-specific genetic and clinical profiles to determine suitability, refine dosing, and track treatment longevity and safety. For instance, in February 2024, the Personalized Medicine Coalition, a US nonprofit organization, noted that the US Food and Drug Administration approved 16 new personalized treatments for rare disease patients in 2023. Therefore, the growing adoption of personalized medicine is a key factor driving the expansion of the hemophilia B gene therapy market.
Which Segment Groups Are Influencing The Hemophilia B Gene Therapy Market?
The hemophilia b gene therapy market covered in this report is segmented –
1) By Therapy: Adeno-Associated Viral (AAV) Vectors; Lentiviral Vectors; Retroviral Vectors
2) By Disease Severity: Severe Hemophilia B; Moderate Hemophilia B; Mild Hemophilia B
3) By Treatment Type: On-Demand; Cure; Prophylaxis
4) By Patient Age Group: Geriatric; Adults; Pediatric
5) By Distribution Channel: Hospital Pharmacies; Specialty Pharmacies
Subsegments:
1) By Adeno-Associated Viral Vectors: Single-Stranded Adeno-Associated Viral Vectors; Self-Complementary Adeno-Associated Viral Vectors; Tissue Specific Promoter Adeno-Associated Viral Vectors; Capsid Modified Adeno-Associated Viral Vectors
2) By Lentiviral Vectors: Integrating Lentiviral Vectors; Non-Integrating Lentiviral Vectors; Self-Inactivating Lentiviral Vectors; Pseudotyped Lentiviral Vectors
3) By Retroviral Vectors: Gamma Retroviral Vectors; Alpha Retroviral Vectors; Moloney Murine Leukemia Virus Retroviral Vectors; Spumaretroviral Vectors
How Are Emerging Trends Affecting The Progression Of The Hemophilia B Gene Therapy Market?
Leading companies active in the hemophilia B gene therapy market are increasingly concentrating on breakthroughs in advanced gene delivery platforms, specifically optimized adeno-associated virus (AAV)–based vector systems, to improve treatment effectiveness and safety. These optimized AAV-based vector systems function as sophisticated viral delivery platforms, enabling the efficient and controlled transfer of functional clotting factor IX genes into target hepatocytes, thereby supporting long-term endogenous factor production. For instance, in May 2023, CSL Behring GmbH, a Germany-based biotechnology company, secured European Commission approval for HEMGENIX, marking it as the first gene therapy sanctioned for the treatment of hemophilia B. HEMGENIX is indicated for adult patients with severe or moderate hemophilia B without a history of factor IX inhibitors and is designed to lessen or eliminate the requirement for routine factor IX prophylaxis. Based on findings from the pivotal HOPE-B clinical study, a significant percentage of treated patients no longer needed regular factor IX infusions, demonstrating enduring therapeutic benefits and emphasizing the potential of next-generation AAV-based gene therapies in long-term disease management.
Who Are The Primary Competitors In The Global Hemophilia B Gene Therapy Market?
Major companies operating in the hemophilia b gene therapy market are REGENXBIO Inc., Pfizer Inc., Bayer Aktiengesellschaft (Bayer AG), Thermo Fisher Scientific Inc., Takeda Pharmaceutical Company Limited, Merck KGaA, CSL Behring LLC, Lonza Group AG, Alnylam Pharmaceuticals Inc., GenScript ProBio LLC, Ultragenyx Pharmaceutical Inc., Takara Bio Inc., Sangamo Therapeutics Inc., Precision BioSciences Inc., Synthego Corporation, MaxCyte Inc., uniQure N.V., Freeline Therapeutics Ltd., Be Biopharma Inc., Asklepios BioPharmaceutical Inc., Cellectis S.A.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/hemophilia-b-gene-therapy-market-report
Which Global Regions Are Shaping The Competitive Landscape Of The Hemophilia B Gene Therapy Market?
North America was the largest region in the hemophilia b gene therapy market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the hemophilia b gene therapy market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Hemophilia B Gene Therapy Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=33162&type=smp
Browse Through More Reports Similar to the Global Hemophilia B Gene Therapy Market 2026, By The Business Research Company
Hepatitis Therapeutics Market Report 2026
https://www.thebusinessresearchcompany.com/report/hepatitis-therapeutics-global-market-report
Acquired Hemophilia Treatment Market Report 2026
https://www.thebusinessresearchcompany.com/report/acquired-hemophilia-treatment-global-market-report
Hemophilia Market Report 2026
https://www.thebusinessresearchcompany.com/report/hemophilia-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model