Respiratory Endotherapy Devices Market Performance Outlook 2026–2030: Revenue to Hit $4.66 Billion at 7.1% CAGR
Uncover key drivers, emerging technologies, and competitive movements shaping the respiratory endotherapy devices market from 2026–2035 with trusted insights from The Business Research Company
What is the expected shift in the Respiratory Endotherapy Devices Market’s size between 2026 and 2030?
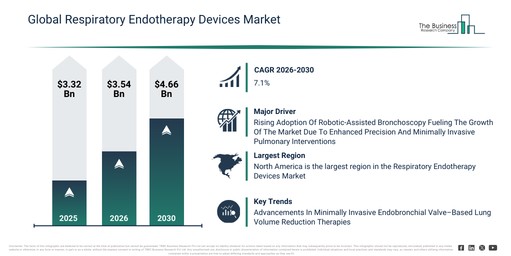
The respiratory endotherapy devices market size has shown significant growth in recent years. It is anticipated to increase from $3.32 billion in 2025 to $3.54 billion in 2026, achieving a compound annual growth rate (CAGR) of 6.9%. The expansion observed historically can be attributed to the rise in chronic respiratory diseases, growth in lung cancer diagnosis, expansion of interventional pulmonology, hospital endoscopy unit growth, and the demand for minimally invasive procedures.
The respiratory endotherapy devices market size is poised for substantial expansion over the coming years, with projections indicating a rise to $4.66 billion by 2030, reflecting a compound annual growth rate (CAGR) of 7.1%. This anticipated growth is driven by several factors, including the increase in early lung screening programs, the adoption of robotic bronchoscopy, the expansion of outpatient pulmonary procedures, the integration of advanced imaging, and the demand for precision airway therapy. Prominent trends throughout the forecast period are expected to encompass minimally invasive airway intervention devices, the widespread use of disposable bronchoscopes, image-guided bronchoscopic procedures, advanced endobronchial ablation systems, and intelligent navigation bronchoscopy tools.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=33296&type=smp
Which Drivers Are Shaping Strategic Decisions In The Respiratory Endotherapy Devices Market?
The increasing integration of robotic-assisted bronchoscopy is expected to drive the growth of the respiratory endotherapy devices market in the coming years. Robotic-assisted bronchoscopy systems represent advanced technology, enabling precise navigation within the respiratory tract and thereby improving diagnostic accuracy and therapeutic outcomes. The demand for these systems is growing as healthcare providers increasingly seek minimally invasive solutions that effectively reduce procedure time, enhance patient safety, and improve overall clinical efficiency. Respiratory endotherapy devices with robotic assistance are crucial for supporting complex interventions by offering real-time imaging capabilities, superior maneuverability, and enhanced control. For instance, in September 2025, as per insights from the European Respiratory Society International Congress, a Switzerland-based membership organization, the utilization of robotic-assisted bronchoscopy coupled with integrated cone beam CT for peripheral lung lesions saw a rise to 84.6% in 2025, significantly up from just 23.1% when using conventional bronchoscopy in 2024. This rising adoption of robotic-assisted bronchoscopy is thus propelling the expansion of the respiratory endotherapy devices market.
What Segment Classifications Make Up The Respiratory Endotherapy Devices Market?
The respiratory endotherapy devices market covered in this report is segmented –
1) By Product Type: Bronchoscopes; Endobronchial Ultrasound Systems; Biopsy Forceps; Guidewires; Catheters; Airway Stents; Endobronchial Valves; Ablation Devices; Other Endotherapy Accessories
2) By Diagnostic Devices: Trans Bronchial Aspiration; Biopsy Forceps; Cytology Brushes; Other Diagnostic Devices
3) By Patient Type: Adult Patients; Pediatric Patients
4) By Specialist: Hospitals; Diagnostic Centers; Ambulatory Surgical Centers
5) By End User: Peripheral Vascular Intervention; Endovascular Interventions; Cardiac; Interventional Radiology; Venous; Other End Users
Subsegments:
1) By Bronchoscopes: Flexible Bronchoscopes; Rigid Bronchoscopes; Video Bronchoscopes; Disposable Bronchoscopes; Therapeutic Bronchoscopes
2) By Endobronchial Ultrasound Systems: Radial Endobronchial Ultrasound Systems; Linear Endobronchial Ultrasound Systems; Portable Endobronchial Ultrasound Systems; Cart Based Endobronchial Ultrasound Systems; High Resolution Endobronchial Ultrasound Systems
3) By Biopsy Forceps: Flexible Biopsy Forceps; Rigid Biopsy Forceps; Disposable Biopsy Forceps; Alligator Jaw Biopsy Forceps; Cup Biopsy Forceps
4) By Guidewires: Hydrophilic Guidewires; Non Coated Guidewires; Steerable Guidewires; Flexible Tip Guidewires; High Strength Guidewires
5) By Catheters: Balloon Catheters; Aspiration Catheters; Delivery Catheters; Diagnostic Catheters; Therapeutic Catheters
6) By Airway Stents: Silicone Airway Stents; Metallic Airway Stents; Covered Airway Stents; Uncovered Airway Stents; Expandable Airway Stents
7) By Endobronchial Valves: One Way Endobronchial Valves; Permanent Endobronchial Valves; Temporary Endobronchial Valves; Adjustable Endobronchial Valves; Implantable Endobronchial Valves
8) By Ablation Devices: Radiofrequency Ablation Devices; Cryoablation Devices; Laser Ablation Devices; Thermal Ablation Devices; Microwave Ablation Devices
9) By Other Endotherapy Accessories: Suction Tubes; Specimen Collection Traps; Bronchoscopy Brushes; Biopsy Needles; Airway Dilators
Which Trends Are Shaping Activity Within The Respiratory Endotherapy Devices Market?
Leading companies in the respiratory endotherapy devices market are focusing on developing advanced solutions, such as endobronchial valves, to minimize treatment risks while achieving targeted lung volume reduction without permanent tissue removal. An endobronchial valve is a minimally invasive implant inserted via bronchoscopy into specific airways to block airflow to diseased lung regions, allowing healthier areas to function more effectively. For instance, in February 2024, Olympus Canada Inc., a Canada-based medical technology company, launched the Spiration Valve System for the treatment of severe emphysema. It is an umbrella-shaped device designed for placement in the most severely diseased lung regions using a minimally invasive bronchoscopic procedure. It uniquely redirects airflow away from hyperinflated lung areas toward healthier tissue, supporting improved respiratory mechanics. As the only endobronchial valve therapy currently available in Canada, it addresses an unmet need in severe emphysema management. Clinical evidence from the EMPROVE trial demonstrates durable benefits through 24 months, including meaningful improvements in lung function, breathlessness, and patient quality of life.
Which Key Market Players Are Investing In Expansion And Innovation Within The Respiratory Endotherapy Devices Market?
Major companies operating in the respiratory endotherapy devices market are Johnson & Johnson, Medtronic plc, Siemens Healthineers, Stryker Corporation, GE Healthcare Technologies Inc., Boston Scientific Corporation, Intuitive Surgical Inc., Olympus Corporation, Hoya Corporation, Steris Healthcare, Teleflex Incorporated, Karl Storz SE & Co. KG, CookMedical, Merit Medical Systems Inc., Ambu A/S, Vygon, Richard Wolf GmbH, Vapotherm Inc, Scholly Fiberoptic GmbH, Clarus Medical LLC, ENDO-FLEX GmbH, The Lung Docs Inc., and Broncus Medical Inc.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/respiratory-endotherapy-devices-market-report
Which Regions Are Poised For Strategic Growth In The Respiratory Endotherapy Devices Market?
North America was the largest region in the respiratory endotherapy devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the respiratory endotherapy devices market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Respiratory Endotherapy Devices Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=33296&type=smp
Browse Through More Reports Similar to the Global Respiratory Endotherapy Devices Market 2026, By The Business Research Company
Respiratory Devices And Equipment Therapeutic Market Report 2026
Endotherapy Devices Market Report 2026
https://www.thebusinessresearchcompany.com/report/endotherapy-devices-global-market-report
Respiratory Devices And Equipment Therapeutic And Diagnostic Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model