Home Spirometry Bluetooth Device Market Outlook 2026–2030 with Major Growth Drivers and Emerging Industry Trends
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
How Much Is The Home Spirometry Bluetooth Device Market Expected To Grow In Terms Of Market Value Between 2026 And 2030?
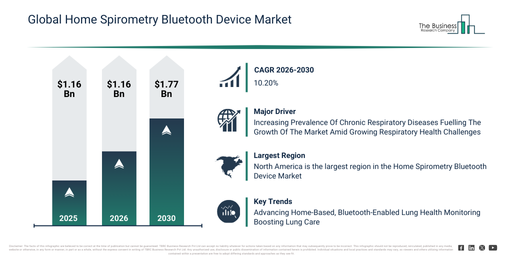
The home spirometry bluetooth device market size has demonstrated significant growth in recent years. It is anticipated to expand from $1.28 billion in 2025 to $1.42 billion in 2026, achieving a compound annual growth rate (CAGR) of 10.5%. This historical expansion can be linked to several factors, including an increase in chronic respiratory diseases, the prompt adoption of home health devices, progress in Bluetooth connectivity, heightened patient self-monitoring awareness, and the integration of these systems with hospitals.
The home spirometry bluetooth device market size is anticipated to undergo significant expansion in the upcoming years. It is projected to attain a value of $2.08 billion by 2030, growing at a compound annual growth rate (CAGR) of 10.2%. The anticipated expansion during this period stems from developments such as AI-powered predictive respiratory analytics, the broadening of remote healthcare programs, integration with telemedicine platforms, increased adoption by homecare providers, and a rising demand for ongoing pulmonary monitoring. Prominent trends for the forecast period encompass wireless pulmonary data transmission, the advent of portable home monitoring devices, real-time tracking of lung function, integration with mobile health applications, and systems for patient-adherence monitoring.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=29083&type=smp
What Drivers Are Influencing The Home Spirometry Bluetooth Device Market?
The anticipated expansion of the home spirometry Bluetooth device market is linked to the rising occurrence of chronic respiratory conditions. These are defined as enduring ailments impacting the lungs’ airways and structures, resulting in diminished lung function and poorer overall respiratory well-being. The primary reason for this increase is the aging demographic, given that elderly individuals are more susceptible to respiratory problems due to natural reductions in lung capacity and prolonged contact with environmental pollutants. Home spirometry Bluetooth devices facilitate continuous remote tracking of lung function for individuals suffering from chronic respiratory diseases, aiding in the prompt identification of deteriorating states for rapid medical intervention and improved sustained care. As an illustration, in May 2025, data from the Centers for Disease Control and Prevention, a US-based government public health agency, indicated that chronic obstructive pulmonary disease (COPD) was the fifth leading cause of death in the United States in 2023, causing 141,733 deaths. During the same year, the age-adjusted prevalence of diagnosed COPD among adults aged 18 years and older stood at 3.8 percent. Consequently, the expanding occurrence of chronic respiratory diseases is fueling the development of the home spirometry Bluetooth device market.
What Major Segment Divisions Exist Within The Home Spirometry Bluetooth Device Market?
The home spirometry bluetooth device market covered in this report is segmented –
1) By Product Type: Handheld Home Spirometry Devices, Compact Tabletop Home Spirometry Devices
2) By Connectivity: Bluetooth-Enabled Devices, Bluetooth + Wi-Fi Enabled Devices
3) By Distribution Channel: Online, Offline
4) By Application: Chronic Obstructive Pulmonary Disease (COPD), Asthma, Cystic Fibrosis, Other Applications
5) By End-User: Homecare Settings, Patients, Hospitals and Clinics, Other End-Users
Subsegments:
1) By Handheld Home Spirometry Devices: Portable Bluetooth Spirometers, Pocket-Size Spirometers, Smartphone-Integrated Spirometers, Rechargeable Handheld Spirometers, Battery-Operated Handheld Spirometers
2) By Compact Tabletop Home Spirometry Devices: Wireless Bluetooth Tabletop Spirometers, Digital Home Diagnostic Spirometers, Multi-Parameter Home Monitoring Spirometers
How Are Trends Shaping The Direction Of The Home Spirometry Bluetooth Device Market?
Leading companies within the home spirometry Bluetooth device market are concentrating on creating advanced solutions, including devices like the Air Next Spirometer, which aids in remote patient oversight and the prompt identification of respiratory ailments such as asthma and COPD. This Air Next Spirometer is a home spirometry instrument that connects via Bluetooth to smartphones, engineered to precisely assess lung function and relay live respiratory data for remote oversight and control. As an illustration, in January 2024, NuvoAir Medical, a US-based firm specializing in tailored heart and lung care, declared that its Air Next Spirometer obtained official 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its deployment as a complete spirometer in home settings. This approval signifies the device is sanctioned for patient use at home, delivering an exhaustive lung function evaluation comparable to assessments performed in healthcare environments. It allows individuals to perform diverse lung function tests, including forced expiratory volume (FEV1) and forced vital capacity (FVC), from their homes, with results forwarded to healthcare providers for continuous management. Furthermore, this clearance facilitates clinical trials, research endeavors, and consistent monitoring of lung health beyond conventional medical facilities, thereby fostering more accessible and uninterrupted respiratory care.
Which Companies Are Leading Innovation In The Home Spirometry Bluetooth Device Market?
Major companies operating in the home spirometry bluetooth device market are Contec Medical Systems Co. Ltd., Vitalograph Ltd., Medcaptain Medical Technology Co. Ltd., Medical International Research S.p.A., CareSimple Inc., Geratherm Medical AG, SDI Diagnostics Inc., NDD Medical Technologies, Futuremed America Inc., Sibelmed S.L., Uscom Ltd., e-LinkCare Meditech Co. Ltd., Monitored Therapeutics Inc. (MTI), Aluna Health Inc., Jones Medical Instrument Company, CMI Health, Healthup SA, Motustech Inc., Medikro Oy, Icen Technology Company Limited
Read the full home spirometry bluetooth device market report here:
Which Regions Are Expected To Experience Rapid Expansion In The Home Spirometry Bluetooth Device Market?
North America was the largest region in the home spirometry bluetooth device market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the home spirometry bluetooth device market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Home Spirometry Bluetooth Device Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=29083&type=smp
Browse Through More Reports Similar to the Global Home Spirometry Bluetooth Device Market 2026, By The Business Research Company
Spirometer Market Report 2026
https://www.thebusinessresearchcompany.com/report/spirometer-global-market-report
Bluetooth Low Energy Ble Technology Market Report 2026
Respiratory Monitoring Devices Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model