Adenosine Deaminase-Severe Combined Immunodeficiency Market Global Report 2026 Market Forecast 2026–2035 Highlighting Market Data Patterns and Industry Insights
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Is The Estimated Market Size Of The Adenosine Deaminase-Severe Combined Immunodeficiency Market During 2026–2030?
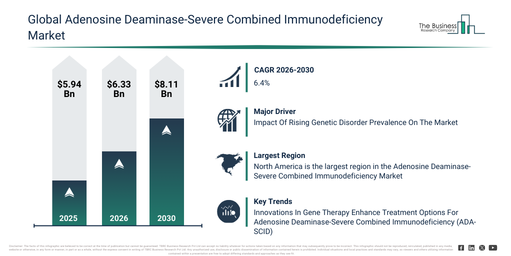
The adenosine deaminase-severe combined immunodeficiency market size has experienced strong expansion in recent years. It is projected to climb from $5.94 billion in 2025 to $6.33 billion in 2026, achieving a compound annual growth rate (CAGR) of 6.6%. The market’s growth during the preceding period can be ascribed to factors such as limited awareness of ADA-SCID, the low availability of gene therapy solutions, a reliance on supportive care treatments, a scarcity of specialized treatment centers, and a high prevalence of treatment delays.
The adenosine deaminase-severe combined immunodeficiency market is projected to experience significant expansion over the coming years. It is anticipated to grow to $8.11 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 6.4%. This projected growth can be attributed to several factors, including advancements in autologous stem cell therapies, improved accessibility to enzyme replacement therapy, an increase in specialized hospitals and clinics, higher rates of pediatric diagnoses, and increased healthcare funding for rare genetic disorders. Dominant trends during the forecast period are expected to include the rising adoption of enzyme replacement therapies, the continued development and enhanced access to gene therapy solutions, an increase in stem cell transplantation procedures, the expansion of pediatric treatment programs for ADA-SCID, and a greater emphasis on supportive care and patient monitoring.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=21085&type=smp
What Underlying Factors Are Accelerating The Growth Of The Adenosine Deaminase-Severe Combined Immunodeficiency Market?
The growing occurrence of genetic disorders is projected to boost the expansion of the adenosine deaminase-severe combined immunodeficiency market in the future. These medical conditions stem from anomalies or mutations in an individual’s DNA, which can be inherited or emerge spontaneously. The increasing incidence of genetic disorders is fueled by elements such as enhanced diagnostic methods, greater public knowledge, older parental age, environmental impacts, and population increase. Specifically, the escalating prevalence of genetic disorders, particularly those like adenosine deaminase-severe combined immunodeficiency (ADA-SCID), stimulates a heightened need for sophisticated diagnostic tools and specific therapeutic approaches. As an illustration, in October 2024, the Cystic Fibrosis Trust, a national charity based in the UK, reported that 11,148 individuals were living with cystic fibrosis (CF) in the UK in 2022, a number that rose to 11,318 by 2023. Consequently, the increasing prevalence of genetic disorders is propelling the growth of the adenosine deaminase-severe combined immunodeficiency market.
How Are The Various Segments Of The Adenosine Deaminase-Severe Combined Immunodeficiency Market Categorized?
The adenosine deaminase-severe combined immunodeficiency market covered in this report is segmented –
1) By Treatment Type: Enzyme Replacement Therapy (ERT), Gene Therapy, Stem Cell Transplantation, Supportive Care Treatments
2) By Route of Administration: Intravenous, Subcutaneous
3) By End-User: Hospitals, Specialty Clinics
Subsegments:
1) By Enzyme Replacement Therapy (ERT): Pegylated Adenosine Deaminase, Non-Pegylated Adenosine Deaminase
2) By Gene Therapy: Autologous Stem Cell-Based Gene Therapy, Viral Vector-Mediated Gene Therapy
3) By Stem Cell Transplantation: HLA-Matched Bone Marrow Transplant, Umbilical Cord Blood Transplant
4) By Supportive Care Treatments: Antimicrobial Therapy, Immune System Support
What Trends Are Affecting The Expansion Of The Adenosine Deaminase-Severe Combined Immunodeficiency Market?
Key organizations operating in the adenosine deaminase-severe combined immunodeficiency treatment market are increasingly focused on developing advanced solutions, such as implementing in-house gene therapy manufacturing, to enhance patient access, reduce costs associated with treatment, and ensure uninterrupted supply. In-house gene therapy production refers to the approach where autologous hematopoietic stem cells, genetically altered with a functional ADA gene, are produced within the organization’s own facilities, thereby lessening reliance on external commercial manufacturers. As an illustration, in February 2025, the Fondazione Telethon, an Italy-based nonprofit biomedical research organization, submitted a Marketing Authorization Application (MAA) to the European Medicines Agency (EMA) for etuvetidigene autotemcel. This gene therapy was initially developed for ADA-SCID and is now also targeting Wiskott-Aldrich Syndrome. The therapy involves a single administration of autologous CD34+ stem and progenitor cells transduced with a lentiviral vector encoding the functional gene, demonstrating promising safety and efficacy results in treated patients.
Which Major Firms Influence Developments In The Adenosine Deaminase-Severe Combined Immunodeficiency Market?
Major companies operating in the adenosine deaminase-severe combined immunodeficiency market are GlaxoSmithKline plc, Chiesi Farmaceutici S.p.A., Orchard Therapeutics PLC, Rocket Pharmaceuticals, Novartis AG, Bluebird Bio, Spark Therapeutics, Leadiant Biosciences, Sigma-Tau Pharmaceuticals, Astellas Pharma, Takeda Pharmaceutical Company, Pfizer Inc, Sanofi, Bristol-Myers Squibb, Roche Holding AG, Johnson & Johnson, AbbVie Inc, Gilead Sciences, CSL Limited, Bayer AG
Read the full adenosine deaminase-severe combined immunodeficiency market report here:
Which Region Is Projected To Dominate The Adenosine Deaminase-Severe Combined Immunodeficiency Market During The Forecast Period?
North America was the largest region in the adenosine deaminase-severe combined immunodeficiency market in 2025. The regions covered in the adenosine deaminase-severe combined immunodeficiency market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Adenosine Deaminase-Severe Combined Immunodeficiency Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=21085&type=smp
Browse Through More Reports Similar to the Global Adenosine Deaminase-Severe Combined Immunodeficiency Market 2026, By The Business Research Company
adeno associated viral vectors global market report
immunoassay global market report
https://www.thebusinessresearchcompany.com/report/immunoassay-global-market-report
Acne Drugs Global Market Report
https://www.thebusinessresearchcompany.com/report/acne-drugs-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model