Artificial Intelligence Clinical Trial Protocol Feasibility Tool Market Revenue Expected to Reach $2.75 Billion by 2030 with 27% CAGR
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Is The Expected Market Size Of The Artificial Intelligence Clinical Trial Protocol Feasibility Tool Market From 2026 To 2030?
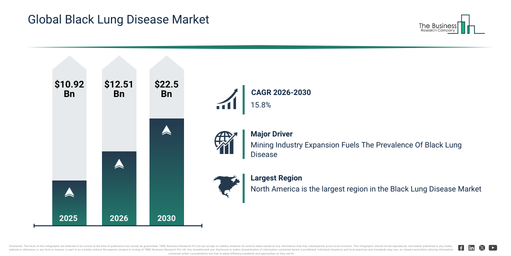
The market size for artificial intelligence clinical trial protocol feasibility tools has experienced rapid expansion in recent times. This market is projected to grow from $0.83 billion in 2025 to reach $1.06 billion in 2026, achieving a compound annual growth rate (CAGR) of 27.3%. The growth observed in the historic period can be attributed to the increasing complexity of protocols in clinical trials, a rise in trial failure rates, a growing reliance on data-driven feasibility analysis, the expansion of global clinical research activities, and the early adoption of AI-based planning tools.
The market size for artificial intelligence clinical trial protocol feasibility tools is poised for significant expansion in the upcoming years. It is projected to achieve a valuation of $2.76 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 27.0%. This anticipated growth is driven by factors such as an escalating need for expedited trial approvals, the increasing embrace of decentralized trial methodologies, the proliferation of AI-powered protocol optimization platforms, a concentrated effort on decreasing trial expenditures, and a heightened focus on predictive feasibility modeling. Notable trends expected throughout the forecast period include the broader application of AI in assessing protocol risks, a surge in the adoption of predictive recruitment modeling, the growing incorporation of historical trial data, the expansion of cloud-based feasibility solutions, and a stronger emphasis on enhancing trial design.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28142&type=smp
What Major Drivers Are Influencing Demand In The Artificial Intelligence Clinical Trial Protocol Feasibility Tool Market?
The increasing focus on precision medicine is anticipated to drive the expansion of the artificial intelligence clinical trial protocol feasibility tool market going forward. Precision medicine describes a personalized healthcare strategy that utilizes a patient’s genetics, environment, and lifestyle to deliver more accurate and effective treatments. The heightened attention on precision medicine is a result of advancements in genomic sequencing and biomarker identification, which enable precise detection and targeted therapies. Artificial intelligence clinical trial protocol feasibility tools enhance precision medicine by facilitating data-driven trial design, rendering them ideal for tailoring treatments to individual patient populations. They mitigate trial inefficiencies by forecasting patient recruitment, timelines, and risks, thereby improving clinical research accuracy and outcomes. For instance, in March 2024, Novotech, an Australia-based biotechnology company, indicated that in 2023, 43% of the 217 FDA-approved oncology therapies were precision oncology treatments, with 78 incorporating DNA or NGS-detectable biomarkers. Consequently, the growing emphasis on precision medicine is fueling the growth of the artificial intelligence clinical trial protocol feasibility tool market.
Which Segments Are Included In The Analysis Of The Artificial Intelligence Clinical Trial Protocol Feasibility Tool Market?
The artificial intelligence clinical trial protocol feasibility tool market covered in this report is segmented –
1) By Component: Software, Services
2) By Deployment Mode: Cloud-Based, On-Premises
3) By Application: Protocol Design, Site Selection, Patient Recruitment, Risk Assessment, Other Applications
4) By End-User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Academic And Research Institutes, Other End-Users
Subsegments:
1) By Software: Predictive Analytics, Machine Learning, Natural Language Processing, Data Integration
2) By Services: Consulting, Implementation, Training And Support, Managed Services
How Are Trends Impacting The Artificial Intelligence Clinical Trial Protocol Feasibility Tool Market?
Leading companies in the artificial intelligence clinical trial protocol feasibility tool market are concentrating on developing innovative solutions, such as predictive analytics-based trial planning software, to improve the accuracy of protocol design and minimize trial delays. This predictive analytics-based trial planning software is a solution that utilizes historical and real-time clinical trial data to forecast timelines, identify potential risks, and optimize study design for more efficient and cost-effective trials. For instance, in June 2024, Lokavant Inc., a US-based clinical intelligence company, introduced Spectrum, the inaugural artificial intelligence clinical trial feasibility solution aimed at optimizing trials from strategic planning through mid-study re-forecasting. The platform features predictive analytics-based trial planning software, enabling continuous, real-time feasibility analysis, timeline and cost prediction, and financial forecasting without manual intervention. Spectrum delivers dynamic, real-time (re)forecasting by integrating live trial data, which allows study teams to monitor progress, explore alternative scenarios, and optimize enrollment outcomes. This solution also provides highly accurate predictions for timelines, costs, and operational risks, supporting faster, data-driven decision-making throughout the clinical trial lifecycle.
Which Key Industry Participants Are Active In The Artificial Intelligence Clinical Trial Protocol Feasibility Tool Market?
Major companies operating in the artificial intelligence clinical trial protocol feasibility tool market are IQVIA Holdings Inc., SAS Institute Inc., Veeva Systems Inc., Tempus AI Inc., Cytel Inc., ArisGlobal LLC., Saama Technologies Inc., Norstella, ConcertAI, Komodo Health Inc., H1 Inc., TriNetX Inc., ObjectiveHealth LLC, Lokavant Inc., Inato Inc., Clinerion Ltd., Faro Health Inc., Lantern Pharma Inc., Ryght AI, BEKHealth Corporation
Read the full artificial intelligence clinical trial protocol feasibility tool market report here:
Which Region Shows The Strongest Potential For Future Growth In The Artificial Intelligence Clinical Trial Protocol Feasibility Tool Market?
North America was the largest region in the artificial intelligence clinical trial protocol feasibility tool market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the artificial intelligence clinical trial protocol feasibility tool market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Artificial Intelligence Clinical Trial Protocol Feasibility Tool Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=28142&type=smp
Browse Through More Reports Similar to the Global Artificial Intelligence Clinical Trial Protocol Feasibility Tool Market 2026, By The Business Research Company
Ai Based Clinical Trial Solution Providers Global Market Report
Ai In Clinical Trials Global Market Report
https://www.thebusinessresearchcompany.com/report/ai-in-clinical-trials-global-market-report
Clinical Trial Management Systems Global Market Report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model