Atrial Septal Defect Market Size Outlook and Demand Growth Across 2026–2030
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Is The Estimated Market Size Of The Atrial Septal Defect Market During 2026–2030?
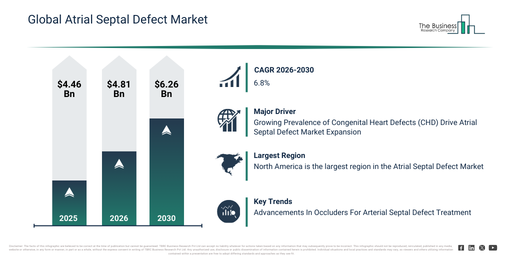
The atrial septal defect market size has experienced significant expansion in recent years. It is anticipated to increase from $4.46 billion in 2025 to $4.81 billion in 2026, reflecting a compound annual growth rate (CAGR) of 7.8%. This historical growth can be attributed to several factors including enhanced detection of congenital heart defects, improved access to surgical correction techniques, the expansion of pediatric cardiology services, accumulating clinical experience with device-based closures, and broader availability of diagnostic imaging technologies.
The atrial septal defect market size is anticipated to experience robust expansion in the coming years. It is projected to reach $6.26 billion by 2030, achieving a compound annual growth rate (CAGR) of 6.8%. This growth during the forecast period is attributable to the increasing embrace of hybrid closure techniques, a heightened focus on early intervention in adults, the expansion of minimally invasive cardiac surgery capabilities, ongoing innovation in occluder device design, and a rising demand for long-term follow-up care solutions. Noteworthy trends expected in the forecast period involve the increasing adoption of transcatheter closure devices, a growing preference for minimally invasive repair procedures, the expanded use of advanced cardiac imaging techniques, the proliferation of pediatric and adult ASD screening programs, and an enhanced emphasis on long-term patient outcomes.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=16345&type=smp
What Underlying Factors Are Accelerating The Growth Of The Atrial Septal Defect Market?
The rising incidence of congenital heart defects (CHD) is projected to boost the atrial septal defect market’s growth. CHD refers to structural heart abnormalities present at birth. The increasing number of people diagnosed with these birth-present structural heart conditions contributes to this trend. Atrial septal defect treatment helps manage CHD by correcting abnormal blood flow between heart chambers, improving heart function, and reducing complications. For instance, in September 2025, the British Heart Foundation, a UK-based non-profit, stated that 2.3 million people in the UK live with congenital heart disease. Moreover, at least 1 in 150 births are diagnosed with these defects, averaging 13 babies daily in the UK, with more diagnoses later. Estimates suggest 1-2 percent of the population may be affected. Thus, the prevalence of congenital heart defects will drive the atrial septal defect market.
How Are The Various Segments Of The Atrial Septal Defect Market Categorized?
The atrial septal defect market covered in this report is segmented –
1) By Treatment Procedure: Surgical Closure, Transcatheter Closure, Hybrid Procedures, Medication Therapy
2) By Diagnosis: Chest X-Ray, Electrocardiogram, Cardiac Catheterization, Transesophageal Echocardiography, Pulse Oximetry
3) By Product Type: Medical Devices, Pharmaceutical Products
4) By Age Group: Pediatric, Adult
5) By End-User: Hospitals, Ambulatory Surgical Centers, Cardiac Clinics, Other End-Users
Subsegments:
1) By Surgical Closure: Open Heart Surgery (Traditional Surgical Closure), Minimally Invasive Surgical Closure, Patches (Pericardial, Synthetic) For Asd Closure
2) By Transcatheter Closure: Device-Based Closure (Amplatzer Septal Occluder), Transcatheter Plug Closure Techniques, Catheter-Based Asd Repair For Adult And Pediatric Patients
3) By Hybrid Procedures: Combination Of Surgical And Transcatheter Techniques, Minimally Invasive Hybrid Procedures, Hybrid Closure For Complex Or Large Asds
4) By Medication Therapy: Anticoagulant Therapy (To Prevent Stroke), Diuretics (For Managing Symptoms), Antihypertensive Drugs (For Managing Associated Conditions), Off-Label Use Of Other Medications
What Key Trends Are Influencing The Development Of The Atrial Septal Defect Market?
Leading companies within the arterial septal defect market are developing advanced devices and seeking regulatory approval to enhance their availability and applications. Device approval refers to obtaining official authorization or certification for a device, typically from a regulatory body, before it can be legally manufactured, sold, or used in a specific market or for a particular purpose. For instance, in March 2024, Occlutech GmbH, a Sweden-based specialist in minimally invasive cardiac devices, announced that the United States Food and Drug Administration (FDA) had approved the Occlutech ASD Occluder and Occlutech Pistol Pusher for treating atrial septal defects (ASD). This approval signifies a major milestone for the company, which has been dedicated to advancing global healthcare. The Occlutech ASD Occluder is designed as a lifelong solution for patients with echocardiography-confirmed defects. It is a self-expanding nitinol device featuring two flexible discs attached to both sides of the patient’s atrial septum using the Occlutech Pistol Pusher. With this approval, Occlutech will initiate commercialization in the US through an exclusive partnership with B. Braun Interventional Systems.
Which Major Firms Influence Developments In The Atrial Septal Defect Market?
Major companies operating in the atrial septal defect market are Abbott Laboratories, Medtronic plc, Boston Scientific Corporation, Edwards Lifesciences Corporation, W. L. Gore & Associates Inc., Cook Medical, Lepu Medical Technology Co. Ltd., MicroPort Scientific Corporation, Lifetech Scientific Corporation, Venus Medtech Hangzhou Inc, Occlutech Holding AG, Osypka AG, Cardia Inc, Gore Medical, Pfm Medical AG, Balton Sp. z o.o., Meril Life Sciences, TTK Healthcare, Braile Biomedica, Shanghai Shape Memory Alloy
Read the full atrial septal defect market report here:
https://www.thebusinessresearchcompany.com/report/atrial-septal-defect-global-market-report
Which Region Is Projected To Dominate The Atrial Septal Defect Market During The Forecast Period?
North America was the largest region in the atrial septal defect market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the atrial septal defect market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Atrial Septal Defect Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=16345&type=smp
Browse Through More Reports Similar to the Global Atrial Septal Defect Market 2026, By The Business Research Company
Left Atrial Appendage Closure Device Global Market Report
Atrial Fibrillation Global Market Report
https://www.thebusinessresearchcompany.com/report/atrial-fibrillation-global-market-report
Congenital Heart Defect Devices Global Market Report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model