Global Infectious Disease Clinical Trials Market Expected to Reach $21.24 Billion by 2030 with 7.1% CAGR
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
How Large Is The Infectious Disease Clinical Trials Market Projected To Become By 2030 Based On Its 2026 Valuation?
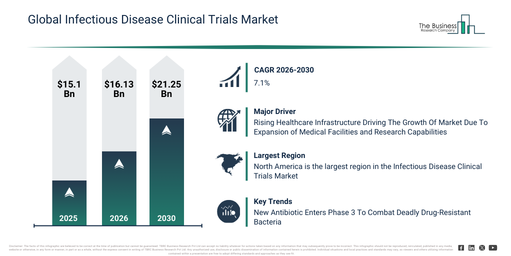
Significant growth has been observed in the infectious disease clinical trials market size recently. Projections indicate it will expand from $15.1 billion in 2025 to $16.13 billion by 2026, demonstrating a compound annual growth rate (CAGR) of 6.8%. Historically, this growth is linked to factors such as an increasing global infectious disease burden, enhanced financial support for vaccine creation, the development of multinational clinical trial networks, the implementation of digital trial management systems, and a rise in public health research efforts.
The infectious disease clinical trials market size is projected to experience robust expansion over the coming years. This market is forecast to reach $21.25 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 7.1%. The anticipated growth during this period is attributed to factors such as heightened investments in pandemic preparedness, the increasing adoption of AI-assisted patient recruitment, wider implementation of global trial decentralization, an intensifying emphasis on accelerated regulatory approvals, and greater integration of digital biomarkers. Key developments expected in the forecast period include a greater embrace of data-driven trial design, the increased utilization of decentralized clinical trial models, deeper integration of real-world evidence, the broadening application of adaptive trial methodologies, and a stronger focus on rapid pathogen response.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=29097&type=smp
What Major Growth Drivers Are Shaping The Outlook Of The Infectious Disease Clinical Trials Market?
The increasing healthcare infrastructure is anticipated to drive the expansion of the infectious disease clinical trials market in the future. Healthcare infrastructure encompasses physical facilities, medical equipment, technologies, and skilled personnel crucial for delivering healthcare services and facilitating effective disease prevention, diagnosis, and treatment. The development of healthcare infrastructure is accelerating due to a higher demand for advanced medical services, the increasing prevalence of new and complex diseases, and the requirement for well-equipped facilities and proficient healthcare professionals to ensure prompt diagnosis, treatment, and preventive care. This infrastructure directly bolsters clinical trial capacity through the provision of cutting-edge medical facilities, specialized equipment, and improved research capabilities vital for executing thorough infectious disease studies. As an illustration, in January 2025, the American Hospital Association, a US-based non-profit organization, forecast that the total number of hospitals in the United States would reach 6,093 by 2025, indicating a slight expansion in healthcare facilities across the nation. Consequently, the expansion of healthcare infrastructure serves as a key driver for the growth of the infectious disease clinical trials market.
What Segment Groups Are Identified Within The Infectious Disease Clinical Trials Market?
The infectious disease clinical trials market covered in this report is segmented –
1) By Phase: Phase I, Phase II, Phase III, Phase IV
2) By Indication: Human Immunodeficiency Virus, Hepatitis, Influenza, Tuberculosis, Malaria, Coronavirus Disease Of 2019 (COVID-19), Other Indications
3) By Study Design: Randomized Controlled Trials, Open Label Studies, Double Blind Studies, Observational Studies, Cohort Studies
4) By Drug Type: Antibiotics, Antivirals, Antifungals, Vaccines, Combination Therapies
5) By End-User: Pharmaceutical Companies, Biotechnology Companies, Contract Research Organizations, Academic Institutes, Other End-Users
Subsegments:
1) By Phase I: Healthy Volunteer Trials, Dose Escalation Studies, Safety And Tolerability Studies, Pharmacokinetic And Pharmacodynamic Studies
2) By Phase II: Early Efficacy Studies, Proof Of Concept Studies, Patient Population Specific Studies, Short Term Safety Studies
3) By Phase III: Large Scale Efficacy Trials, Comparative Effectiveness Studies, Multi Center Clinical Trials, Extended Safety And Tolerability Studies
4) By Phase IV: Post Marketing Surveillance Studies, Long Term Safety Studies, Real World Evidence Studies, Special Population Studies
What Key Trends Are Influencing The Development Of The Infectious Disease Clinical Trials Market?
Major companies operating in the infectious disease clinical trials market are concentrating on developing sophisticated research initiatives, such as phase 3 trials for antibiotic candidates, to assist in discovering rare side effects and establishing optimal dosing. A phase 3 trial for antibiotic candidates is a late-stage investigation that tests the drug in a substantial patient group to evaluate its safety, effectiveness, and comparison to existing treatments before it can be approved for use. For instance, in May 2025, Roche, a Switzerland-based pharmaceutical company, commenced a Phase 3 clinical trial for zosurabalpin, a novel tethered macrocyclic peptide antibiotic specifically engineered to combat CRAB, a critical contributor to severe hospital infections worldwide. Zosurabalpin’s distinctive characteristic lies in its novel mechanism of action, which impedes the transport of lipopolysaccharide essential for building the bacterial outer membrane, with no resistance observed to date. This global trial will encompass approximately 400 patients to assess its safety and effectiveness when contrasted with standard treatments. Preliminary research has demonstrated powerful activity against multidrug-resistant bacteria, excellent tolerability, and encouraging pharmacokinetic properties. This positions Zosurabalpin as a potentially revolutionary solution in tackling the urgent challenge of antibiotic resistance.
Which Companies Hold Significant Positions In The Infectious Disease Clinical Trials Market?
Major companies operating in the infectious disease clinical trials market are GlaxoSmithKline plc (GSK), Pfizer Inc, Moderna Inc, Novartis AG, Sanofi S.A., AstraZeneca plc, Merck & Co Inc, Gilead Sciences Inc, AbbVie Inc, Eli Lilly and Company, Bristol Myers Squibb Company, Roche Holding AG, Bayer AG, Takeda Pharmaceutical Company Limited, CSL Limited, Regeneron Pharmaceuticals Inc, Biogen Inc, Amgen Inc, Inovio Pharmaceuticals Inc, Dynavax Technologies Corporation, Valneva SE, Emergent BioSolutions Inc, Vir Biotechnology Inc, International AIDS Vaccine Initiative, VBI Vaccines Inc, Otsuka Pharmaceutical Co Ltd, Daiichi Sankyo Company Limited, Shionogi & Co Ltd, Astellas Pharma Inc, Eisai Co Ltd, Sun Pharmaceutical Industries Ltd, Glenmark Pharmaceuticals Ltd, Bharat Biotech International Limited, Laurus Labs Limited, Sarepta Therapeutics Inc, Alnylam Pharmaceuticals Inc, Intellia Therapeutics Inc, Sangamo Therapeutics Inc, Excision BioTherapeutics Inc, Atara Biotherapeutics Inc, Adaptive Biotechnologies Corporation, ImmunityBio Inc, Enanta Pharmaceuticals Inc, Iterum Therapeutics plc, Spero Therapeutics Inc, Venatorx Pharmaceuticals Inc, Vaxart Inc, Immunovant Inc, Tonix Pharmaceuticals Holding Corp, Genentech Infectious Diseases Unit
Read the full infectious disease clinical trials market report here:
Which Regions Are Projected To Dominate The Infectious Disease Clinical Trials Market In The Coming Years?
North America was the largest region in the infectious disease clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the infectious disease clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Infectious Disease Clinical Trials Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=29097&type=smp
Browse Through More Reports Similar to the Global Infectious Disease Clinical Trials Market 2026, By The Business Research Company
Hospital Acquired Infection Control Global Market Report
Infection Prevention Global Market Report
https://www.thebusinessresearchcompany.com/report/infection-prevention-global-market-report
Infection Control Supplies Global Market Report
https://www.thebusinessresearchcompany.com/report/infection-control-supplies-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model