Renal Toxicity ADME Testing Market Set to Grow to $2.78 Billion by 2030 at 12.9% CAGR
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Is The Forecasted Market Value Of The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Across 2026–2030?
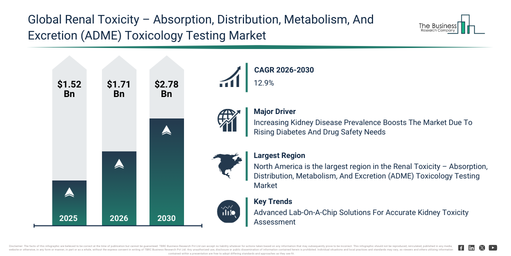
The market for renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing has seen rapid expansion in recent years. It is anticipated to grow from $1.52 billion in 2025 to $1.71 billion in 2026, at a compound annual growth rate (CAGR) of 12.7%. The reasons for this past growth include increased preclinical drug safety testing, a rise in drug failures related to nephrotoxicity, the growth of CRO services, more stringent regulatory safety requirements, and greater adoption of in vitro assays.
The renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market size is anticipated to experience swift expansion over the upcoming years. It is projected to reach $2.78 billion in 2030, demonstrating a compound annual growth rate (CAGR) of 12.9%. This projected growth can be attributed to an increase in biologics and complex drugs, the wider adoption of predictive toxicology models, the continued expansion of organ-on-chip platforms, advancements in precision medicine research, and the rising demand for AI-based safety analytics. Significant trends expected during the forecast period include kidney organoid-based toxicity assays, AI-assisted nephrotoxicity prediction models, high-throughput renal screening platforms, biomarker-driven renal safety panels, and microphysiological kidney-on-chip systems.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=33295&type=smp
What Key Drivers Are Influencing The Growth Of The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market?
The increasing occurrence of kidney-related conditions is projected to propel the expansion of the renal toxicity-absorption, distribution, metabolism, and excretion (ADME) toxicology testing market going forward. These disorders are health issues that damage the kidneys, affecting their capacity to filter blood and maintain body balance. The rise in kidney-related disorders stems from growing rates of diabetes, which harms kidney blood vessels and impairs their filtering capabilities. Renal toxicity ADME/toxicology testing aids in detecting and evaluating potential kidney damage induced by drugs or chemicals, thereby enabling early intervention and the development of safer treatments. For instance, according to the American Cancer Society, a US-based professional organization, approximately 80,980 new cases of kidney (renal) cancer are expected to be diagnosed in the US in 2025, with an estimated 14,510 deaths resulting from the disease. Therefore, the escalating prevalence of kidney-related disorders is driving the growth of the renal toxicity-absorption, distribution, metabolism, and excretion (ADME) toxicology testing market.
What Segmentation Levels Are Considered In The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market?
The renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market covered in this report is segmented –
1) By Test Type: In Vitro Renal Toxicity Testing; In Vivo Renal Toxicity Testing; In Silico Renal Toxicity Modeling
2) By Method: Biochemical and Enzymatic Assays; Cell Based Renal Assays; Biomarker Based Toxicity Analysis; Histopathology and Imaging; Pharmacokinetic and Drug Metabolism Studies
3) By Stage of Drug Development: Discovery and Lead Optimization; Preclinical Development; Clinical Development; Post Marketing Safety Studies
4) By Technology: High Throughput Screening Platforms; Organ On Chip and Microfluidic Systems; Omics Based Toxicology Platforms; Artificial Intelligence and Predictive Modeling Tools; Digital Pathology and Imaging Systems
5) By End User: Pharmaceutical and Biotechnology Companies; Contract Research Organizations; Academic and Research Institutes; Regulatory Laboratories
Subsegments:
1) By In Vitro Renal Toxicity Testing: Primary Renal Cell Assays; Immortalized Renal Cell Line Assays; Three Dimensional Renal Cell Cultures; Kidney Organoid Models; Transporter Interaction Assays; Biomarker Based Nephrotoxicity Assays; High Content Imaging Assays; Microphysiological Kidney System Models
2) By In Vivo Renal Toxicity Testing: Acute Renal Toxicity Studies; Subacute Renal Toxicity Studies; Subchronic Renal Toxicity Studies; Chronic Renal Toxicity Studies; Renal Biomarker Evaluation Studies; Histopathological Kidney Assessment; Dose Response Toxicity Studies; Recovery and Reversibility Studies
3) By In Silico Renal Toxicity Modeling: Quantitative Structure Activity Relationship Modeling; Machine Learning Based Toxicity Prediction; Physiologically Based Pharmacokinetic Modeling; Virtual Screening for Nephrotoxicity; Read Across Toxicity Modeling; Systems Biology Based Toxicity Modeling
What Trends Are Projected To Affect The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market?
Leading companies within the renal toxicity-absorption, distribution, metabolism, and excretion (ADME) toxicology testing market are concentrating on developing novel solutions, such as low-drug-absorbing microfluidic chips, to enhance the predictive accuracy of kidney toxicity while decreasing reliance on animal studies. A low-drug-absorbing microfluidic chip is a lab-on-a-chip apparatus designed for renal toxicity assessment that minimizes unintended drug absorption by its material, ensuring a more precise simulation of kidney functions and reliable measurement of drug effects on renal cells. For instance, in September 2024, Emulate Inc., a US-based biotechnology company, introduced the chip-R1 rigid chip to improve kidney toxicity testing, yielding more accurate and dependable results. This chip is engineered for precise ADME and toxicology modeling in non-stretch organs like the liver and kidney, utilizing low-drug-absorbing plastics and a thin, porous polycarbonate membrane to prevent compound loss and enhance drug recovery for lipophilic compounds. Its advantages include high shear stress support (up to 2.3 dyn/cm²), preactivated surfaces for simpler workflows, and improved cellular crosstalk, while its benefits encompass better reproducibility, accurate toxicity detection, and compatibility with existing organ-chip hardware to accelerate human-relevant preclinical testing.
Who Are The Dominant Players Shaping The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Landscape?
Major companies operating in the renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market are Thermo Fisher Scientific Inc., Eurofins Scientific SE, Agilent Technologies Inc., WuXi AppTec Co. Ltd., Charles River Laboratories International Inc., PerkinElmer Inc., Beckman Coulter Inc., Bio-Rad Laboratories Inc., Pharmaron Beijing Co. Ltd., Simulations Plus Inc., GVK Biosciences Pvt. Ltd., Promega Corporation, Syngene International Ltd., SAI Life Sciences Ltd., QPS Holdings LLC, Southern Research Institute, BioIVT LLC, TCG Lifesciences Pvt. Ltd., Frontage Laboratories Inc., Symeres, TSRL Inc., BOC Sciences LLC, Vivisciences Preclinical CRO.
Read the full renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market report here:
Which Regions Are Projected To Dominate The Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market In The Coming Years?
North America was the largest region in the renal toxicity-ADME toxicology testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the renal toxicity – absorption, distribution, metabolism, and excretion (adme) toxicology testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=33295&type=smp
Browse Through More Reports Similar to the Global Renal Toxicity – Absorption, Distribution, Metabolism, And Excretion (ADME) Toxicology Testing Market 2026, By The Business Research Company
Adme Toxicology Testing Market Report 2026
https://www.thebusinessresearchcompany.com/report/adme-toxicology-testing-global-market-report
Renal Biomarkers Market Report 2026
https://www.thebusinessresearchcompany.com/report/renal-biomarkers-global-market-report
Kidney Dialysis Market Report 2026
https://www.thebusinessresearchcompany.com/report/kidney-dialysis-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model