Clinical Trial Supplies Market Expansion Forecast Reaching $4.32 Billion By 2030 At 8.7% CAGR
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
How Is The Market Size Of The Clinical Trial Supplies Market Expected To Change From 2026 To 2030?
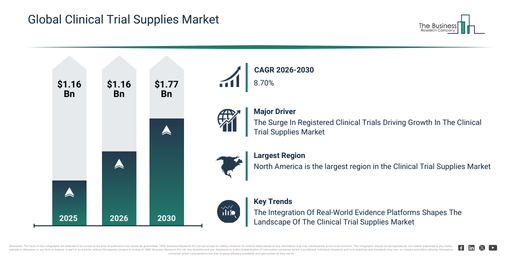
The clinical trial supplies market has experienced substantial growth in recent years. It is projected to expand from $2.86 billion in 2025 to $3.1 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 8.3%. This historical expansion can be linked to an increase in global clinical trial activity, the wider adoption of multicenter trial designs, the growth of biologics and specialty drugs, a rise in outsourcing to CROs, and the improvement of cold-chain logistics infrastructure.
The clinical trial supplies market size is projected for significant expansion in the coming years, with its value anticipated to reach $4.33 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 8.7%. This growth during the forecast period is primarily driven by factors such as the increasing complexity of clinical trial protocols, the expansion of decentralized and virtual trials, a rising focus on patient-centric trial models, growing investments in digital trial management tools, and increasing regulatory scrutiny across regions. Major trends identified for the forecast period include the increasing adoption of integrated supply chain platforms, the rising use of temperature-controlled logistics solutions, a growing demand for end-to-end trial visibility, the expansion of adaptive packaging and labeling services, and an enhanced focus on compliance and traceability.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12133&type=smp
What Key Factors Are Shaping The Clinical Trial Supplies Market Landscape?
The expanding volume of registered clinical trials is anticipated to boost the clinical trial supplies market in the coming period. These studies involve human subjects to assess the safety and efficacy of novel medical treatments, interventions, or diagnostic methods. Necessary for successful trials, these supplies, which encompass pharmaceuticals, medical devices, and other vital items, are key to supporting research, guaranteeing precise data collection, and safeguarding patient well-being. An illustration of this trend is provided by ClinicalTrials.gov, a branch of the National Institutes of Health (NIH), a US government entity that oversees and funds medical research; in May 2023, it reported approximately 437,533 registered clinical trials in 2023, an increase from 399,499 registered studies in 2022, spanning all 50 states of the United States and 221 countries. Consequently, the rising number of registered clinical trials serves as a primary driver for the expansion of the clinical trial supplies market.
How Are Different Segments Classified In The Clinical Trial Supplies Market Segment Analysis?
The clinical trial supplies market covered in this report is segmented –
1) By Services: Logistics And Distribution, Storage And Retention, Supply Chain Management, Packaging, Labeling, And Blinding, Comparator Sourcing
2) By Clinical Phases: Phase I, Phase II, Phase III, Phase IV
3) By Therapeutic Use: Oncology, Central Nervous System (CNS), Cardiovascular, Infectious Disease, Metabolic Disorders, Other Therapeutic Uses
4) By End User: Pharmaceutical And Biotech companies, Contract Research Organization (CRO)
Subsegments:
1) By Logistics And Distribution: Transportation, Inventory Management, Temperature-Controlled Shipping, Customs Clearance
2) By Storage And Retention: Short-term Storage, Long-term Storage, Controlled Environment Storage
3) By Supply Chain Management: Planning And Forecasting, Supplier Management, Risk Management, Compliance Management
4) By Packaging, Labeling, And Blinding: Primary Packaging, Secondary Packaging, Label Design And Printing, Blinding Techniques
5) By Comparator Sourcing: Sourcing Of Reference Products, Quality Control And Assurance, Regulatory Compliance For Comparators
What Market Trends Are Affecting The Clinical Trial Supplies Market?
Major entities within the clinical trial supplies market are implementing advanced technologies to revolutionize healthcare decision-making, drug development processes, and patient support. They achieve this by utilizing real-world data to generate evidence-based findings and maintain their competitive standing. As an illustration, in April 2023, Komodo Health, a healthcare technology firm based in the US specializing in patient recruitment, site selection, and real-time data monitoring for clinical trials, introduced MapEnhance. This innovation represents a network of specialized data partners integrated into a real-world evidence (RWE) technology platform. MapEnhance provides clients with an extensive and granular real-world perspective on clinical events and patient demographics, facilitating unparalleled insights. Its features encompass precision molecular diagnostics across various therapeutic domains, high-volume standard laboratory diagnostics for earlier disease identification, and electronic medical records that capture clinical interactions. Furthermore, it incorporates inpatient and outpatient facility chargemasters, offering detailed insights into costs and utilization across diverse hospital environments.
Who Are The Companies Driving Activity In The Clinical Trial Supplies Market?
Major companies operating in the clinical trial supplies market are Thermo Fisher Scientific Inc., IQVIA, Eurofins Scientific SE, Parexel International Corporation, ICON PLC, Catalent Inc., Intertek Group PLC, Recipharm AB, World Courier, Almac Group Ltd., Piramal Pharma Solutions, Clinigen Group PLC, Movianto GmbH, Marken Limited, PCI Pharma Services, Rubicon Research Private Limited, Bionical Ltd., Durbin PLC, SIRO Clinpharm Pvt. Ltd., Biocair International Ltd., Ancillare LP., Myonex, Klifo A/S, Alium Medical Limited, ADAllen Pharma, Sharp Services LLC
Get The Full Clinical Trial Supplies Market Report:
https://www.thebusinessresearchcompany.com/report/clinical-trial-supplies-global-market-report
Which Region Is The Largest In The Clinical Trial Supplies Market?
North America was the largest region in the clinical trial supplies market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the clinical trial supplies market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Clinical Trial Supplies Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/customise?id=12133&type=smp
Browse Through More Reports Similar to the Global Clinical Trial Supplies Market 2026, By The Business Research Company
Medical And Diagnostic Laboratory Services Market Report 2026
Medical And Diagnostic Laboratory Services Market Report 2026
Veterinary Reference Laboratory Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model