Rising Adoption of Minimally Invasive Procedures Boosts Congenital Heart Defect Device Sales Is Supporting Expansion In The Congenital Heart Defect Devices Market
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Level Of Market Value Growth Is Predicted For The Congenital Heart Defect Devices Market From 2026 To 2030?
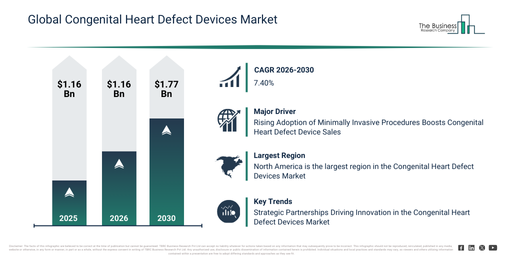
The congenital heart defect devices market has experienced robust expansion recently. This market is projected to expand from $3.11 billion in 2025 to $3.34 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 7.4%. Historically, this growth has been driven by factors such as the increasing occurrence of congenital heart defects, significant progress in pediatric cardiac surgical techniques, improved survival rates for patients with congenital heart conditions, the establishment of more specialized cardiac care facilities, and enhanced recognition of early congenital defect diagnosis.
The congenital heart defect devices market is anticipated to show significant expansion in the next few years, with its size forecast to reach $4.45 billion in 2030, exhibiting a compound annual growth rate (CAGR) of 7.4%. This growth during the forecast period is primarily attributed to an increased emphasis on early diagnosis and intervention, the rising demand for minimally invasive pediatric cardiac treatments, improved access to advanced cardiac care within emerging markets, a growing population of adults with congenital heart disease, and continuous innovation in implantable cardiac devices. Major trends expected in this period include the increasing adoption of minimally invasive congenital heart procedures, greater utilization of transcatheter and implantable cardiac devices, a heightened focus on early-life and pediatric cardiac interventions, the expansion of device-based therapies for complex heart defects, and an escalating demand for long-term cardiac monitoring and support devices.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=18543&type=smp
Which Primary Drivers Are Impacting The Congenital Heart Defect Devices Market Growth?
The escalating adoption of minimally invasive procedures is projected to fuel the expansion of the congenital heart defect device market. These procedures entail surgical operations conducted through minor cuts or natural orifices, frequently employing specialized instruments and methods to lessen injury and improve recuperation. The rise in minimally invasive procedures stems from technological progress, patients’ inclination for less intrusive therapies, enhanced surgical methods, and the advantages of quicker recovery and fewer complications. Congenital heart defect devices are integral to minimally invasive procedures, aiding in the identification, care, and control of cardiac irregularities via approaches that use smaller incisions or catheter-based techniques. For example, in June 2024, data from the American Society of Plastic Surgeons, a US-based organization, indicated that in 2023, plastic surgeries grew by 5%, while minimally invasive procedures saw a 7% increase over the previous year. Consequently, the growing frequency of minimally invasive procedures is propelling the congenital heart defect devices market.
Which Segment Classifications Are Used In The Congenital Heart Defect Devices Market Segment Analysis?
The congenital heart defect devices market covered in this report is segmented –
1) By Device Type: Catheters, Pacemakers, Single-Chamber Pacemakers, Dual-Chamber Pacemakers, Biventricular Pacemakers, Leadless Pacemakers, Implantable Cardioverter Defibrillators, Biventricular Devices, Implanted Cardiac Loop Recorders, Other Device Types
2) By Defect Type: Atrial Septal Defect, Ventricular Septal Defect, Atrioventricular Septal Defect, Tricuspid Atresia, Truncus Arteriosus, Other Defect Types
3) By End user: Hospitals, Specialty Clinics, Academic And Research Institute, Other End Users
Subsegments:
1) By Catheters: Balloon Catheters, Diagnostic Catheters, Guiding Catheters
2) By Pacemakers: Single-Chamber Pacemakers, Dual-Chamber Pacemakers, Biventricular Pacemakers, Leadless Pacemakers
3) By Implantable Cardioverter Defibrillators: Single-Chamber Icds, Dual-Chamber Icds, Subcutaneous Icds
4) By Biventricular Devices: Crt-P (Cardiac Resynchronization Therapy Pacemakers), Crt-D (Cardiac Resynchronization Therapy Defibrillators)
5) By Implanted Cardiac Loop Recorders: Traditional Implantable Loop Recorders, Subcutaneous Implantable Loop Recorders
6) By Other Device Types: Transcatheter Heart Valves, Ventricular Assist Devices (VADS)
Which Trends Are Shaping Growth In The Congenital Heart Defect Devices Market?
Leading companies in the congenital heart defect devices market are adopting strategic partnerships to develop innovative solutions for congenital heart defects. These alliances support businesses in the congenital heart defect devices sector by pooling resources and expertise, accelerating research and development, broadening market access, and speeding up commercialization. Ultimately, this collaboration fosters innovation and drives growth within the industry. For instance, in May 2024, SMT (Sahajanand Medical Technologies Pvt Ltd), an India-based manufacturing company, partnered with HeartX, an India-based MedTech firm, to enhance patient care in the cardiovascular sector. Through this partnership, Sahajanand Medical Technologies (SMT) strengthens its position in the congenital heart defect (CHD) space by integrating HeartX’s innovative solutions, such as the JOVE VB Stent. This strategic alliance enables SMT to offer advanced CHD devices that enhance treatment efficacy, simplify procedures, and improve patient outcomes.
Which Firms Are Contributing To The Congenital Heart Defect Devices Market Ecosystem?
Major companies operating in the congenital heart defect devices market are Johnson & Johnson, Abbott Laboratories, Medtronic plc, Boston Scientific Corporation, W L Gore & Associates Inc, Lepu Medical Technology Beijing Co Ltd, MicroPort Scientific Corporation, Berlin Heart GmbH, Syncardia Systems LLC, Occlutech Holding AG, Sahajanand Medical Technologies Pvt Ltd, Xeltis AG, Procyrion Inc, Heartstitch Inc, Cardia Inc, OSYPKA Medical GmbH, On-X Life Technologies Inc, Ventracor Limited, HemoFlow Technologies, NuPulseCV Inc
Get The Full Congenital Heart Defect Devices Market Report:
Which Region Is The Top Contributor To The Congenital Heart Defect Devices Market By Share?
North America was the largest region in the congenital heart defect devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the congenital heart defect devices market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Congenital Heart Defect Devices Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/customise?id=18543&type=smp
Browse Through More Reports Similar to the Global Congenital Heart Defect Devices Market 2026, By The Business Research Company
Computed Tomography Ct Scanners Devices And Equipment Market Report 2026
Computed Tomography Ct Scanner Market Report 2026
Positron Emission Tomography Pet Scanners Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model