Global Non-Radiographic Axial Spondyloarthritis Therapeutic Market 2026–2030: Forecast Insights for Business Planning
Uncover key drivers, emerging technologies, and competitive movements shaping the non-radiographic axial spondyloarthritis therapeutic market from 2026–2035 with trusted insights from The Business Research Company
Starting from its 2026 valuation, what market size is the Non-Radiographic Axial Spondyloarthritis Therapeutic Market expected to reach by 2030?
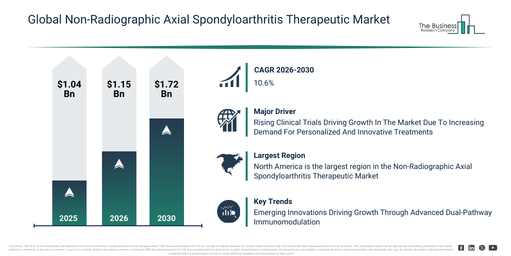
The non-radiographic axial spondyloarthritis therapeutic market has experienced substantial expansion in recent times. Its valuation is projected to increase from $1.04 billion in 2025 to $1.15 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 10.9%. This historical growth can be linked to factors such as a heightened awareness of non-radiographic axial spondyloarthritis, the shortcomings of traditional NSAID treatments, the implementation of better diagnostic standards, broader access to rheumatology specialized care, and a wider availability of biologic medications.
The market for therapies addressing non-radiographic axial spondyloarthritis is projected to experience swift expansion over the coming years, with its size anticipated to reach $1.72 billion by 2030, reflecting a compound annual growth rate (CAGR) of 10.6%. This growth during the forecast period is primarily driven by the accelerated development of next-generation biologics, the increasing adoption of JAK inhibitor therapies, a growing focus on personalized rheumatology care, the expansion of early-stage intervention protocols, and improvements in patient awareness and diagnosis rates. Major trends expected within this period include the rising adoption of biologic disease-modifying therapies, increased utilization of IL-17 and TNF inhibitors, a stronger emphasis on early disease intervention, the expansion of targeted small molecule treatments, and an enhanced focus on long-term disease control.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=24627&type=smp
What Drivers Are Driving Adoption Within The Non-Radiographic Axial Spondyloarthritis Therapeutic Market?
The non-radiographic axial spondyloarthritis therapeutic market is anticipated to expand, driven by the rising number of clinical trials. These trials involve human research to assess the safety, efficacy, and adverse reactions of medical therapies or interventions. The rise in clinical trials stems from a heightened need for tailored and novel treatments, which necessitate thorough safety and effectiveness evaluations. For non-radiographic axial spondyloarthritis therapeutics, clinical trials offer evidence-based confirmation of treatment effectiveness and safety during early disease stages, facilitating regulatory acceptance and widespread clinical use. An example of this trend is seen in the UK, where the Association of the British Pharmaceutical Industry, a government and NHS organization focused on enhancing patient access to new treatments, stated in December 2024 that the total number of industry-initiated clinical trials increased from 411 trials in 2022 to 426 in 2023. Consequently, the expansion of clinical trials is a key factor boosting the non-radiographic axial spondyloarthritis therapeutic market.
How Is The Non-Radiographic Axial Spondyloarthritis Therapeutic Market Organized Into Various Segments?
The non-radiographic axial spondyloarthritis therapeutic market covered in this report is segmented –
1) By Type: Non-Steroidal Anti-Inflammatory Drugs, Biologics, Small Molecule Therapies, Corticosteroids
2) By Application: Oral, Injectable, Parenteral
3) By End User: Hospitals, Ambulatory Surgical Centers, Homecare Settings
Subsegments:
1) By Non-Steroidal Anti-Inflammatory Drugs: COX-2 Inhibitors, Non-Selective NSAIDs
2) By Biologics: Tumor Necrosis Factor (TNF) Inhibitors, Interleukin-17 (IL-17) Inhibitors
3) By Small Molecule Therapies: Janus Kinase (JAK) Inhibitors, TYK2 Inhibitors
4) By Corticosteroids: Oral Corticosteroids, Intra-Articular Corticosteroid Injections
Which Trends Are Guiding The Evolution Of The Non-Radiographic Axial Spondyloarthritis Therapeutic Market?
Leading firms within the non-radiographic axial spondyloarthritis (nr-axSpA) therapeutic market are prioritizing the creation of novel approaches like dual IL-17A/IL-17F biologic inhibitors and highly convenient delivery methods. This focus responds to increasing diagnosis rates, earlier disease identification, the necessity for swift and profound inflammatory control, and patient preference for less frequent or more accessible dosing. Dual IL-17A/IL-17F biologic inhibitors consist of monoclonal antibodies that specifically and concurrently inhibit the cytokines IL-17A and IL-17F, which contributes to the management of nr-axSpA by alleviating inflammation, pain, stiffness, and structural progression. As an illustration, in September 2024, UCB S.A. (Union Chimique Belge Société Anonyme), a Belgium-based global biopharmaceutical company, introduced BIMZELX in the U.S. for adults experiencing active nr-axSpA, following FDA approval. BIMZELX is a humanized IgG monoclonal antibody engineered to neutralize both IL-17A and IL-17F, offering more extensive suppression of IL-17-mediated pathways compared with conventional TNF inhibitors or single-target IL-17 treatments. Beyond its use in nr-axSpA, BIMZELX also holds indications for moderate-to-severe plaque psoriasis, psoriatic arthritis, and ankylosing spondylitis, thereby catering to the changing therapeutic requirements of patients with inflammatory conditions.
Who Are The Primary Competitors In The Global Non-Radiographic Axial Spondyloarthritis Therapeutic Market?
Major companies operating in the non-radiographic axial spondyloarthritis therapeutic market are Pfizer Inc, Johnson and Johnson, AbbVie Inc, Sanofi SA, Bristol Myers Squibb Company, AstraZeneca PLC, Novartis AG, Eli Lilly and Company, Gilead Sciences Inc, Amgen Inc, Biogen Inc, Daiichi Sankyo Company Limited, UCB SA, Mochida Pharmaceutical Co Ltd, Mereo BioPharma Group plc, Suzhou Zelgen Biopharmaceuticals Co Ltd, Qyuns Therapeutics Co Ltd, Sandoz Group AG, Boehringer Ingelheim GmbH, Roche Holding AG
Access The Complete Report For Deeper Market Insights:
Which Regions Are Poised For Strategic Growth In The Non-Radiographic Axial Spondyloarthritis Therapeutic Market?
North America was the largest region in the non-radiographic axial spondyloarthritis therapeutics market in 2025. The regions covered in the non-radiographic axial spondyloarthritis therapeutic market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Non-Radiographic Axial Spondyloarthritis Therapeutic Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=24627&type=smp
Browse Through More Reports Similar to the Global Non-Radiographic Axial Spondyloarthritis Therapeutic Market 2026, By The Business Research Company
Ankylosing Spondylitis Market Report 2026
https://www.thebusinessresearchcompany.com/report/ankylosing-spondylitis-global-market-report
Cervical Spondylosis Treatment Market Report 2026
Osteoarthritis Market Report 2026
https://www.thebusinessresearchcompany.com/report/osteoarthritis-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model