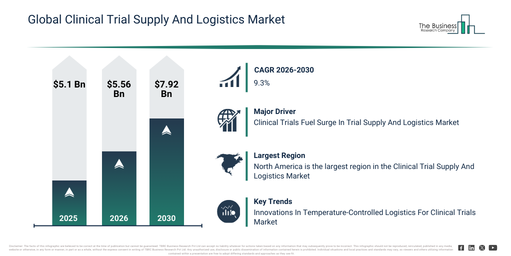
Clinical Trial Supply & Logistics Market to Reach $7.92B by 2030 at 9.3% CAGR
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Expansion In Market Value Is Forecasted For The Clinical Trial Supply And Logistics Market Between 2026 And 2030?
The clinical trial supply and logistics market has seen substantial growth in recent years. This market is anticipated to expand from $5.1 billion in 2025 to $5.56 billion in 2026, achieving a compound annual growth rate (CAGR) of 8.9%. The historical increase in size can be linked to a rising number of global clinical trials, the broader adoption of multinational study designs, the increasing complexity of trial protocols, a greater demand for temperature-controlled logistics, and enhanced outsourcing of trial supply operations.
The clinical trial supply and logistics market is anticipated to experience substantial expansion in the coming years. Projections indicate it will reach $7.92 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 9.3%. This future growth is primarily driven by factors such as the wider uptake of decentralized clinical trials, a growing need for patient-focused supply models, the expansion of trials involving biologics and cell therapies, increasing financial commitments to digital trial logistics platforms, and a heightened emphasis on monitoring supply in real-time. Key developments expected during this period involve the greater integration of cold chain logistics, a heightened requirement for comprehensive trial supply visibility, the increasing implementation of centralized inventory management systems, the growth of direct-to-patient supply approaches, and a stronger emphasis on reducing risks and ensuring continuous supply.
Access Your Free Sample Report For In-Depth Market Analysis:
Which Factors Are Influencing The Growth Of The Clinical Trial Supply And Logistics Market?
A rising volume of clinical trials is anticipated to drive the future expansion of the clinical trial supply and logistics market. Clinical trials involve research conducted with human participants to assess the safety and efficacy of new medical treatments, interventions, or diagnostic procedures. Clinical trial supply and logistics services are essential for providing necessary materials, such as the collection and delivery of biological specimens, investigational drugs and kits, along with expert support for all permit applications and customs clearance, which ensures the smooth execution of these research studies. For example, in July 2023, data from ClinicalTrials.gov, a U.S.-based registry of clinical trials operated by the United States National Library of Medicine at the National Institutes of Health (NIH), showed that the number of registered clinical studies climbed from 399,499 in 2022 to 437,533 by July of 2023 across all 50 states of the United States and 221 countries. It was also reported that 141,513 studies (31% of the total) were registered in the U.S. only, while 244,707 studies (53% of the total) were registered in non-U.S. locations. Thus, the increasing prevalence of clinical trials is a significant driver for the growth of the clinical trial supply and logistics market.
What Are The Key Segment Divisions In The Clinical Trial Supply And Logistics Market Segment Structure?
The clinical trial supply and logistics market covered in this report is segmented –
1) By Service: Logistics And Distribution, Storage And Retention, Packaging Labelling And Blinding, Manufacturing, Comparator Sourcing, Other Services
2) By Phase: Phase I, Phase II, Phase III, Phase IV
3) By Area: Oncology, Cardiovascular Diseases, Respiratory Diseases, CNS And Mental Disorders, Other Areas
4) By End-User: Pharmaceuticals, Biologicals, Medical Device
Subsegments:
1) By Logistics And Distribution: Transportation Management, Cold Chain Logistics, Customs Clearance, Inventory Management
2) By Storage And Retention: Controlled Storage, Long-Term Storage, Short-Term Storage
3) By Packaging Labeling, And Blinding: Primary Packaging, Secondary Packaging, Label Design And Printing, Blinding Solutions
4) By Manufacturing: Clinical Supply Manufacturing, Batch Production, Quality Control And Assurance
5) By Comparator Sourcing: Sourcing Services, Regulatory Compliance, Inventory Management For Comparators
6) By Other Services: Regulatory Affairs Consulting, Clinical Trial Management, Data Management And Analysis, Training And Support Services
Which Trends Are Influencing Demand In The Clinical Trial Supply And Logistics Market?
Major companies in the Clinical Trial Supply and Logistics Market are concentrating on technologies such as temperature-controlled healthcare shipments to enhance product integrity and reliability during transport, ensuring compliance with regulatory standards and improving patient outcomes. These shipments refer to the transportation of medical products, pharmaceuticals, and biological materials that require specific temperature conditions to maintain their efficacy and safety, utilizing specialized packaging, monitoring systems, and logistics solutions to keep products within the required temperature range throughout transit. For instance, in February 2024, FedEx Corporation, a US-based transport company, launched its FedEx Life Science Center, including FedEx Customized Freight (FCF), for transporting critical pharmaceutical shipments. This service features highest boarding priority, end-to-end custodial control, and dedicated delivery options tailored to preserve product integrity during transit. Additionally, customers can utilize FedEx Priority Alert Plus, which offers proactive monitoring of time-sensitive shipments with options for replenishment of dry ice or gel packs.
Which Organizations Are Engaged In The Clinical Trial Supply And Logistics Market?
Major companies operating in the clinical trial supply and logistics market are United Parcel Service, DHL Group, FedEx Corporation, Thermo Fisher Scientific Inc., IQVIA, ICON plc, Eurofins Scientific, Catalent Inc., Intertek Group plc, Parexel International Corporation, UDG Healthcare PLC., Almac Group, World Courier, Piramal Pharma Solutions, Sharp Clinical Services Ltd., Clinigen Group, PCI Pharma Services, Movianto Group, Cold Chain Technologies, Biocair International Limited, QuickSTAT, Wasdell Group, Klifo A/S, TrakCel, Packaging Coordinators Inc., Biotec Services International Limited, Proventa International, Inceptua Group
Get The Full Clinical Trial Supply And Logistics Market Report:
Which Region Leads The Clinical Trial Supply And Logistics Market In Terms Of Market Share?
North America was the largest region in the clinical trial supply and logistics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the clinical trial supply and logistics market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Clinical Trial Supply And Logistics Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Clinical Trial Supply And Logistics Market 2026, By The Business Research Company
Clinical Trial Management System Market Report 2026
Clinical Trial Supplies Market Report 2026
https://www.thebusinessresearchcompany.com/report/clinical-trial-supplies-global-market-report
Clinical Trial Management Systems Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model