eTMF systems market forecast with demand insights
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Level Of Market Value Growth Is Predicted For The Electronic Trial Master File (eTMF) Systems Market From 2026 To 2030?
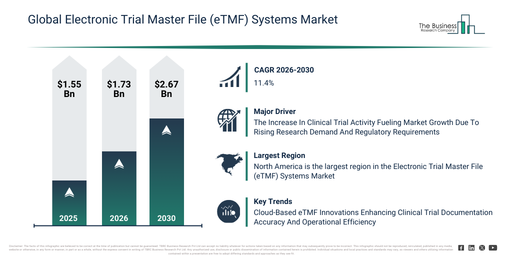
The electronic trial master file (etmf) systems market has seen considerable growth in recent years. Its valuation is expected to rise from $1.55 billion in 2025 to $1.73 billion in 2026, at a compound annual growth rate (CAGR) of 11.8%. Key drivers contributing to this growth in previous periods include regulatory documentation requirements, the increasing complexity of clinical trials, the inherent limitations of paper-based TMFs, the necessity for audit readiness, and the expansion of trials globally.
The electronic trial master file (etmf) systems market size is projected to experience swift expansion in the upcoming years. This market is predicted to reach $2.67 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 11.4%. Factors contributing to this growth over the forecast period include decentralized trial growth, ai-driven document classification, cloud adoption in clinical research, regulatory scrutiny increase, and a stronger focus on data security. Key trends anticipated during the forecast period encompass centralized clinical document management, cloud-based etmf platforms, regulatory compliance automation, real-time trial documentation tracking, and secure data archiving systems.
Access Your Free Sample Report For In-Depth Market Analysis:
Which Strong Drivers Are Impacting The Electronic Trial Master File (eTMF) Systems Market Growth?
The increasing quantity of clinical trials is anticipated to drive the expansion of the electronic trial master file (eTMF) systems market in the coming period. These trials are structured research studies carried out to assess the safety, efficacy, and overall impact of medical interventions like drugs, therapies, and medical devices within specific patient populations. The escalation in clinical trial activities stems from rising global healthcare research and development investments, expanding therapeutic innovations, and more stringent regulatory demands requiring precise, compliant, and accessible study documentation. Electronic trial master file (eTMF) systems facilitate the clinical research ecosystem by digitalizing trial documentation, enabling real-time collaboration, ensuring readiness for audits, and enhancing operational efficiency across various trial sites. For instance, in December 2024, as per the Association of the British Pharmaceutical Industry (ABPI), a UK-based trade association, the count of industry-sponsored clinical trials launched in the UK rose from 411 in 2022 to 426 in 2023. Thus, the growing number of clinical trials is fueling the enlargement of the electronic trial master file (eTMF) systems market.
How Are Different Segments Classified In The Electronic Trial Master File (eTMF) Systems Market Segment Analysis?
The electronic trial master file (etmf) systems market covered in this report is segmented –
1) By Component: Services, Software
2) By Delivery Mode: On-Premise, Cloud-Based
3) By End-User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations (CROs), Other End-Users
Subsegments:
1) By Services: Consulting Services, Implementation Services, Training And Support Services, Data Migration Services
2) By Software: eTMF Management Software, Document Management Software, Cloud-Based eTMF Solutions, Compliance And Regulatory Software
Which Trends Are Contributing To Changes In The Electronic Trial Master File (eTMF) Systems Market?
Leading companies in the electronic trial master file (eTMF) systems market are concentrating on developing sophisticated cloud-based platforms to elevate document precision, strengthen audit preparedness, and optimize clinical trial workflows. These cloud-based eTMF systems are defined as digital, cloud-hosted solutions specifically engineered to securely and efficiently oversee crucial clinical trial documentation, simultaneously facilitating real-time cooperative efforts among dispersed global study teams. As an illustration, during March 2023, Phlexglobal Ltd., an entity within PharmaLex, a Germany-based provider of regulatory and compliance services, introduced PhlexTMF v21. This next-generation, cloud-based eTMF solution integrates advanced artificial intelligence (AI) capabilities. This particular platform is designed to minimize metadata inaccuracies and prevent the misplacement of documents at the initial upload stage, thereby enhancing TMF quality, boosting inspection readiness, and elevating overall trial efficiency. Consequently, advancements in cloud-based eTMF solutions are profoundly transforming clinical documentation management practices and accelerating their widespread adoption throughout the life sciences industry.
Who Are The Companies Driving Activity In The Electronic Trial Master File (eTMF) Systems Market?
Major companies operating in the electronic trial master file (etmf) systems market are Veeva Systems, Oracle, TransPerfect, Phlexglobal, SureClinical Inc., MasterControl Inc, Clinevo Technologies, Covance Inc, Ennov, ePharma Solutions, Aris Global LLC, Montrium Inc, IQVIA, Labcorp Drug Development, Freyr, Medidata (Dassault Systèmes), BSI Life Sciences, OmniComm Systems Inc, Signant Health Ltd, Cloudbyz, Florence Healthcare, Egnyte Inc, Trial Interactive
Get The Full Electronic Trial Master File (eTMF) Systems Market Report:
Which Region Is The Top Contributor To The Electronic Trial Master File (eTMF) Systems Market By Share?
North America was the largest region in the electronic trial master file (eTMF) systems market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the electronic trial master file (etmf) systems market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Electronic Trial Master File (eTMF) Systems Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Electronic Trial Master File (eTMF) Systems Market 2026, By The Business Research Company
Clinical Trial Management System Market Report 2026
Etl Software Market Report 2026
https://www.thebusinessresearchcompany.com/report/etl-software-global-market-report
Clinical Trial Management System Ctms Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model