Episodic ataxia treatment market analysis with growth outlook
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Expansion In Market Value Is Forecasted For The Episodic Ataxia Treatment Market Between 2026 And 2030?
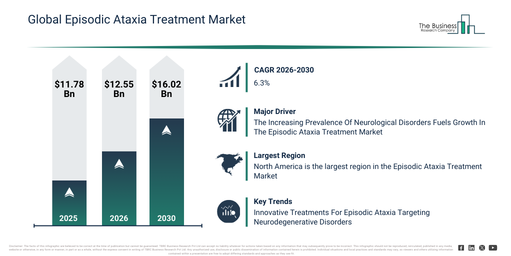
The episodic ataxia treatment market has experienced substantial expansion in recent years. It is anticipated to increase from $11.78 billion in 2025 to $12.55 billion in 2026, reflecting a compound annual growth rate (CAGR) of 6.5%. Historically, this growth can be ascribed to factors such as limited available treatment options, heightened neurologist awareness, improved rare disease diagnosis rates, medication-focused symptom management, and established hospital-based neurology care.
The episodic ataxia treatment market is projected for substantial expansion in the coming years. Its valuation is anticipated to reach $16.02 billion by 2030, driven by a compound annual growth rate (CAGR) of 6.3%. This expansion over the forecast period is primarily due to progress in genetic research, the creation of orphan drugs, tailored neurology treatments, the broadening of homecare options, and enhancements in early diagnosis. Key developments anticipated during this timeframe encompass a heightened emphasis on uncommon neurological conditions, a greater reliance on medications for symptom management, the extension of long-term disease care strategies, an uptick in the implementation of individualized treatment programs, and a strong focus on supportive and rehabilitation services.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/report/episodic-ataxia-treatment-global-market-report
What Key Factors Are Shaping The Episodic Ataxia Treatment Market Landscape?
The expansion of the episodic ataxia treatment market is anticipated to be fueled by the rising incidence of neurological disorders. These disorders encompass illnesses affecting the central and peripheral nervous systems, including elements like the brain, spinal cord, cranial and peripheral nerves, the autonomic nervous system, nerve roots, the neuromuscular junction, and muscles. The heightened occurrence of neurological disorders stems from various factors such as an aging population, genetic predispositions, and infectious diseases. Episodic ataxia treatments are employed in these conditions to alleviate symptoms and enhance the management of intermittent coordination and balance issues. For example, the Alzheimer’s Association, a US-based non-profit organization, noted in April 2024 that in 2023, over 11 million family members and other unpaid caregivers dedicated an estimated 18.4 billion hours to caring for individuals with Alzheimer’s or other dementias. This figure could reach 13.8 million by 2060, unless medical breakthroughs emerge to prevent or cure Alzheimer’s disease. Consequently, the growing prevalence of neurological disorders is a significant driver for the episodic ataxia treatment market.
Which Segment Types Are Examined In The Episodic Ataxia Treatment Market Segment Study?
The episodic ataxia treatment market covered in this report is segmented –
1) By Type: Ataxia Telangiectasia, Episodic Ataxia, Spinocerebellar Ataxia, Friedreich’s Ataxia, Other Types
2) By Treatment: Medications, Levodopa, Pramipexole, Venlafaxine, Other Treatments
3) By Route Of Administration: Oral, Parenteral, Other Routes Of Administrations
4) By Distribution Channel: Direct Tender, Retail Sales, Other Distribution Channels
5) By End User: Hospital, Clinics, Home Healthcare, Other End Users
Subsegments:
1) By Ataxia Telangiectasia: Classic Ataxia Telangiectasia, Variant Ataxia Telangiectasia, Early-Onset Ataxia Telangiectasia, Late-Onset Ataxia Telangiectasia
2) By Episodic Ataxia: Episodic Ataxia Type 1 (EA1), Episodic Ataxia Type 2 (EA2), Episodic Ataxia Type 3 (EA3), Other Episodic Ataxias
3) By Spinocerebellar Ataxia: SCA Type 1, SCA Type 2, SCA Type 3, SCA Type 6
4) By Friedreich’s Ataxia: Classic Friedreich’s Ataxia, Late-Onset Friedreich’s Ataxia, Atypical Friedreich’s Ataxia
5) By Other Types: Secondary Ataxias, Metabolic Ataxias, Drug-Induced Ataxias, Trauma-Induced Ataxias
What Market Trends Are Affecting The Episodic Ataxia Treatment Market?
Leading companies in the episodic ataxia treatment market are concentrating on developing innovative remedies, such as Friedreich’s ataxia (FA) treatment, to improve patient outcomes and address the unmet clinical needs linked to this uncommon neurological condition. Episodic ataxia (EA) is a rare neurological disorder distinguished by intermittent occurrences of ataxia and an inability to voluntarily coordinate muscle movements. These episodes can be short or lengthy and fluctuate in both how often they occur and their severity. For instance, in February 2024, Biogen Inc., a US-based biotechnology firm, announced the approval of SKYCLARYS (omaveloxolone) specifically for treating Friedreich’s ataxia (FA). Omaveloxolone targets the core mitochondrial dysfunction associated with FA, offering the potential to modify the disease’s advancement rather than just providing symptom relief. The therapeutic agent acts as a powerful transcriptional modulator, aiding in the recovery of mitochondrial function and cellular energy creation.
Which Players Are Part Of The Competitive Landscape Of The Episodic Ataxia Treatment Market?
Major companies operating in the episodic ataxia treatment market are BioMarin Pharmaceutical Inc., Neurocrine Biosciences Inc., PTC Therapeutics Inc., Catalyst Pharmaceuticals Inc., Design Therapeutics Inc., Larimar Therapeutics Inc., Voyager Therapeutics Inc., Capsida Biotherapeutics Inc., Adverum Biotechnologies Inc., CRISPR Therapeutics AG, Banner Health, Biogen Inc., Reata Pharmaceuticals Inc., Minoryx Therapeutics, EryDel, Matrix Biomed, IntraBio, Biohaven Pharmaceuticals Ltd., Stealth BioTherapeutics Inc., Acasti Pharma, Seelos Therapeutics Inc., Q-State Biosciences, Locanabio, Lexeo Therapeutics, Retrotope Inc., Ionis Pharmaceuticals Inc.
Get The Full Episodic Ataxia Treatment Market Report:
https://www.thebusinessresearchcompany.com/report/episodic-ataxia-treatment-global-market-report
Where Is The Episodic Ataxia Treatment Market Most Concentrated Geographically?
North America was the largest region in the episodic ataxia treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the episodic ataxia treatment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Episodic Ataxia Treatment Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/episodic-ataxia-treatment-global-market-report
Browse Through More Reports Similar to the Global Episodic Ataxia Treatment Market 2026, By The Business Research Company
Dyslexia Treatment Market Report 2026
https://www.thebusinessresearchcompany.com/report/dyslexia-treatment-global-market-report
Ataxia Market Report 2026
https://www.thebusinessresearchcompany.com/report/ataxia-global-market-report
Progressive Ataxia And Weakness Disorders Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model