Medical Devices Vigilance Market Outlook Highlighting Emerging Trends Influencing Growth
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
By 2030, What Market Size Is The Medical Devices Vigilance Market Expected To Reach Based On Its 2026 Value?
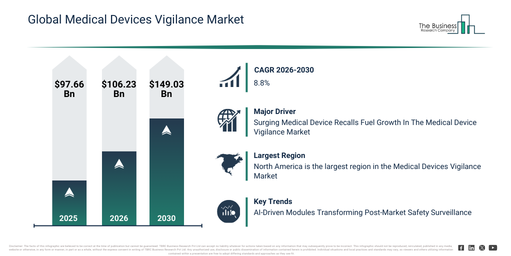
The medical devices vigilance market size has seen substantial growth over recent years. It is anticipated to expand from $97.66 billion in 2025 to $106.23 billion in 2026, reflecting a compound annual growth rate (CAGR) of 8.8%. The historical growth in this sector is largely attributed to an increase in reported device-related adverse events, the implementation of post-market surveillance regulations, a general rise in medical device usage, growing awareness of patient safety, and the expansion of global regulatory frameworks.
The medical devices vigilance market is projected to experience robust expansion in the coming years. Its value is anticipated to reach $149.03 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 8.8%. This projected growth is driven by factors such as the proliferation of connected and intricate medical devices, more stringent post-market compliance demands, an increase in cross-border device authorizations, the growing embrace of proactive risk management strategies, and the need for ongoing safety surveillance. Key trends anticipated during this period encompass the tightening of post-market surveillance mandates, an escalating emphasis on gathering real-world evidence, heightened regulatory oversight concerning adverse event reporting, the broadening of global vigilance standardization efforts, and an increasing need for more rapid incident response systems.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=14137&type=smp
Which Important Drivers Are Guiding The Medical Devices Vigilance Market Growth?
The rising count of medical recalls is anticipated to propel the medical device vigilance market forward. Medical recalls pertain to actions initiated by regulatory bodies, manufacturers, or distributors to withdraw or correct medical products that pose a risk to public health or fail to comply with regulatory standards. Medical device vigilance involves the ongoing monitoring of medical devices in the market to detect adverse events, malfunctions, or safety concerns. Upon detection, these are reported to regulatory authorities, potentially leading to further investigation and, if necessary, a product recall. For instance, in March 2024, Sedgwick, a UK-based risk management and brand protection company, reported that 2023 saw 3,306 medical device recalls in Europe (EU + UK), representing a 20% increase compared to 2022. Therefore, the growing number of medical recalls is indeed driving the expansion of the medical device vigilance market.
What Are The Different Segment Types In The Medical Devices Vigilance Market Segment Breakdown?
The medical devices vigilance market covered in this report is segmented –
1) By Delivery Mode: On-Demand, On-Premise
2) By Application: Therapeutics, Diagnostics, Surgical, Research, Other Applications
3) By End-User: Original Equipment Manufacturers (OEMs), Clinical Research Organizations (CROs), Business Process Outsourcing (BPO) Firms
Subsegments:
1) By On-Demand: Cloud-Based Solutions, Subscription Services, Remote Monitoring And Reporting
2) By On-Premise: In-House Software Solutions, Local Server Deployments, Customized System Implementations
What Trends Are Influencing The Evolution Of The Medical Devices Vigilance Market?
Major companies operating in the medical device vigilance market are prioritizing innovative approaches to post-market safety surveillance, such as artificial intelligence (AI)-driven modules, to automate adverse event reporting, enhance operational efficiency, and improve signal detection. Artificial intelligence (AI)-driven modules refer to intelligent software components that utilize artificial intelligence to process, analyze, and prioritize vigilance data for ensuring regulatory compliance and patient safety. For instance, in July 2024, AB Cube, a France-based medical software company, launched an enhanced customer vigilance platform to support the post-market monitoring of medical devices. The platform integrates adverse event reporting, trend analysis, and compliance documentation into a unified interface; it functions by automating the collection, classification, and reporting of device-related incidents to regulatory authorities. Unique features include real-time analytics dashboards, automated regulatory submissions, and customizable alerts for critical safety events; its applications extend to medical device manufacturers, notified bodies, and hospitals. Benefits include improved patient safety, reduced regulatory risk, faster incident response, and enhanced overall vigilance efficiency.
Who Are The Well-Known Companies In The Medical Devices Vigilance Market?
Major companies operating in the medical devices vigilance market are Johnson & Johnson, Intel Corporation, Oracle Corporation, Medtronic, Siemens Healthineers, RELX Group plc, MasterControl Inc., Laerdal Medical, Numerix, Smithers, Omnify Software Inc., Freyr, Sparta Systems, MDI Consultants Inc., Greenlight Guru, Jama Software, Sarjen Systems Pvt. Ltd, Arena Solutions Inc., Xybion Corporation, ZEINCRO Group, Extedo Gmbh, AssurX Inc., AB-Cube, Panacea Pharma Projects Limited, Qvigilance, General Electric (GE) Healthcare, Philips Healthcare, Baxter International, Stryker Corporation, Boston Scientific
Get The Full Medical Devices Vigilance Market Report:
https://www.thebusinessresearchcompany.com/report/medical-devices-vigilance-global-market-report
Where Is The Medical Devices Vigilance Market Most Concentrated Geographically?
North America was the largest region in the medical devices vigilance market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the medical devices vigilance market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Medical Devices Vigilance Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/medical-devices-vigilance-global-market-report
Browse Through More Reports Similar to the Global Medical Devices Vigilance Market 2026, By The Business Research Company
Healthcare Reimbursement Market Report 2026
https://www.thebusinessresearchcompany.com/report/healthcare-reimbursement-global-market-report
Medical Device Reprocessing Market Report
https://www.thebusinessresearchcompany.com/report/medical-device-reprocessing-global-market-report
Medical Device Outsourcing Market Report
https://www.thebusinessresearchcompany.com/report/medical-device-outsourcing-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model