Viral Vector Purification Market Outlook for 2026–2030 Covering Key Growth Drivers and Industry Trends
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Is The Forecasted Market Size Trajectory Of The Viral Vector Purification Market From 2026 To 2030?
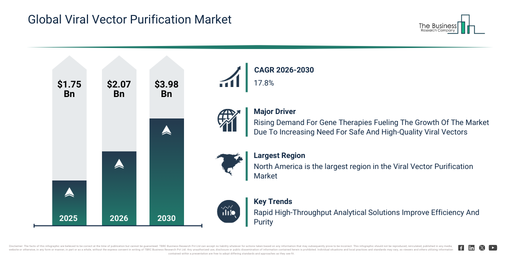
The viral vector purification market has expanded significantly in terms of size recently. This market is projected to expand from $1.75 billion in 2025 to $2.07 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 18.1%. Historically, this expansion was driven by several factors, including an increase in viral vector manufacturing activities, greater uptake of gene therapy research, the initial implementation of ultrafiltration and chromatography methods, a heightened emphasis on vector safety, and the readily available purification reagents and kits.
The viral vector purification market is anticipated to experience significant expansion in the coming years. This market is projected to reach $3.98 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 17.8%. Factors contributing to this growth during the forecast period include a heightened demand for clinical-grade viral vectors, increased investments in downstream bioprocessing technologies, the broadening of gene therapy pipelines, wider adoption of automated purification platforms, and stricter regulatory oversight concerning product purity. Significant trends expected within this period encompass the growing integration of high-resolution chromatography techniques, an escalating need for scalable purification solutions, increased utilization of single-use purification systems, the development of high-purity vector isolation processes, and a stronger emphasis on optimizing process yields.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=30429&type=smp
What Key Factors Are Shaping The Viral Vector Purification Market Landscape?
The increasing need for gene therapies is anticipated to drive the expansion of the viral vector purification market in the future. Gene therapies constitute medical interventions focusing on the insertion, removal, or modification of genetic material within a patient’s cells to manage or avert illnesses. The uptick in demand for gene therapies stems from innovations in delivery systems, which facilitate the secure, effective, and precise conveyance of genetic material into cells. Viral vector purification improves gene therapies by guaranteeing the production of high-quality, safe, and biologically active viral vectors. This process boosts therapy efficacy through the elimination of contaminants and the optimization of vector function, thereby fostering dependable and effective treatment results. For example, data from December 2024, provided by the American Society of Gene & Cell Therapy (ASGCT), a US-based non-profit medical and scientific entity, shows that the Food and Drug Administration (FDA) authorized six gene therapy products in 2023, an increase from five approvals in 2022. Consequently, the escalating need for gene therapies is propelling the expansion of the viral vector purification market.
What Are The Main Segments Within The Viral Vector Purification Market Segment Structure?
The viral vector purification market covered in this report is segmented –
1) By Vector Type: Adenoviral Vectors, Lentiviral Vectors, Adeno-Associated Viral Vectors, Retroviral Vectors, Other Vector Types
2) By Product Type: Kits, Reagents, Instruments, Services
3) By Purification Technique: Chromatography, Ultrafiltration, Precipitation, Other Purification Techniques
4) By Application: Gene Therapy, Vaccinology, Research, Other Applications
5) By End-User: Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Contract Research Organizations, Other End-Users
Subsegments:
1) By Adenoviral Vectors: First Generation, Second Generation, Third Generation
2) By Lentiviral Vectors: Integrating Lentiviral Vectors, Non Integrating Lentiviral Vectors
3) By Adeno Associated Viral Vectors: Recombinant Adeno Associated Viral Vectors, Self Complementary Adeno Associated Viral Vectors
4) By Retroviral Vectors: Gamma Retroviral Vectors, Alpha Retroviral Vectors, Beta Retroviral Vectors
What Market Trends Are Affecting The Viral Vector Purification Market?
Major companies operating in the viral vector purification market are increasingly concentrating on rapid, high-throughput analytical solutions, specifically macro mass photometry platforms, to improve process efficiency, ensure vector purity, and shorten development timelines. Macro mass photometry platforms are defined as instruments that utilize light scattering to determine both the size and mass of viral particles, thereby enabling precise evaluation of empty versus full capsids and the detection of degraded particles in minutes. For example, in November 2023, Refeyn Ltd., a UK-based biotechnology company, launched KaritroMP, a macro mass photometry platform, which conducts analysis of adenovirus and lentivirus vectors within six minutes. The platform provides semi-automated sample processing for up to 13 samples, delivers strong reproducibility for quality control, and permits the identification of subtle differences in particle mass to support purification optimization. This introduction enhances the capabilities of viral vector manufacturers by offering faster and more precise assessment of vector quality. It also aids in streamlining workflow efficiency, consequently reducing the time and resources required for vector characterization and purification.
Who Are The Companies Participating In The Viral Vector Purification Market?
Major companies operating in the viral vector purification market are Merck KGaA, Thermo Fisher Scientific Inc., Lonza Group AG, FUJIFILM Diosynth Biotechnologies, Danaher Corporation, Sartorius AG, Agilent Technologies Inc., Eurofins Scientific SE, Charles River Laboratories International Inc., Bio-Rad Laboratories Inc., AGC Biologics A/S, Takara Bio Inc., Miltenyi Biotec B.V. & Co. KG, Oxford Biomedica PLC, Revvity Inc., Applied Biological Materials Inc. (ABM), Creative Biolabs Inc., Repligen Corporation, Asahi Kasei Corporation, Promega Corporation, 3M Company
Get The Full Viral Vector Purification Market Report:
https://www.thebusinessresearchcompany.com/report/global-viral-vector-purification-market-report
Which Geographic Region Dominates The Viral Vector Purification Market?
North America was the largest region in the viral vector purification market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the viral vector purification market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Viral Vector Purification Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/global-viral-vector-purification-market-report
Browse Through More Reports Similar to the Global Viral Vector Purification Market 2026, By The Business Research Company
Viral Vector Manufacturing Market Report 2026
https://www.thebusinessresearchcompany.com/report/viral-vector-manufacturing-global-market-report
Viral And Non Viral Vector Manufacturing Market Report
Viral Inactivation Market Report
https://www.thebusinessresearchcompany.com/report/viral-inactivation-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model