Viral Vector-Based Gene Therapy Market Growth Analysis Highlighting Innovation and Competitive Landscape
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Is The Estimated Market Size Of The Viral Vector-Based Gene Therapy Market In 2026, And How Will Its Value Evolve By 2030?
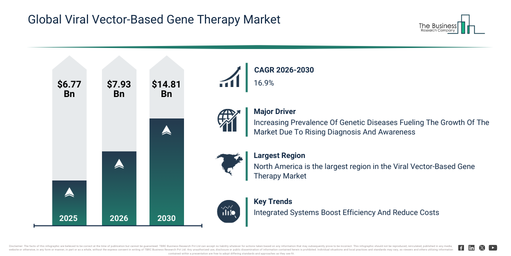
The viral vector-based gene therapy market has experienced significant expansion in its size over recent years. It is projected to increase from $6.77 billion in 2025 to $7.93 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 17.2%. The growth observed in the past can be attributed to advancements in molecular biology research, a deeper understanding of genetic disorders, successful initial clinical applications of viral vector therapies, the expansion of orphan drug programs, and the accessibility of viral vector platforms.
The viral vector-based gene therapy market is projected to experience substantial expansion in the coming years. By 2030, this market is anticipated to reach a valuation of $14.81 billion, progressing at a compound annual growth rate (CAGR) of 16.9%. This projected growth is driven by factors such as the increasing number of approved gene therapy products, a surge in investments for rare disease treatments, the broadening scope of precision medicine initiatives, the expanding acceptance of personalized healthcare, and ongoing advancements in vector engineering technologies. Key trends anticipated during this period involve the increasing uptake of targeted gene therapies, the evolving development of personalized treatment strategies, an increase in clinical trials for rare diseases, a heightened focus on the long-term effectiveness of therapies, and a stronger emphasis on vector safety and overall performance.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=30431&type=smp
What Factors Are Contributing To The Growth Of The Viral Vector-Based Gene Therapy Market?
The rising occurrence of genetic diseases is projected to propel the expansion of the viral vector-based gene therapy market in the upcoming period. Genetic diseases, which are conditions caused by DNA changes or mutations that can be inherited or emerge spontaneously, affect health, development, or physiological functions. The increasing prevalence of these diseases is attributable to heightened awareness and advancements in diagnostic technologies, enabling earlier and more accurate detection of previously undiagnosed conditions. Viral vector-based gene therapy improves the management of genetic diseases by offering targeted delivery of therapeutic genes, rendering it ideal for treating inherited disorders. It mitigates disease progression through precise and enduring correction of genetic defects, thereby enhancing patient outcomes and quality of life. For instance, in April 2025, the International Diabetes Federation (IDF), a Belgium-based non-profit organization, stated that approximately 589 million people were living with diabetes in 2024, and this total is forecast to increase to 853 million by 2050. Therefore, the growing prevalence of genetic diseases is stimulating the growth of the viral vector-based gene therapy market.
Which Segments Define The Viral Vector-Based Gene Therapy Market Segment Structure?
The viral vector-based gene therapy market covered in this report is segmented –
1) By Vector Type: Adenoviral Vectors, Adeno-Associated Viral Vectors, Lentiviral Vectors, Retroviral Vectors
2) By Delivery Method: Intravenous Injection, Intrathecal Injection, Intramuscular Injection, Subcutaneous Injection, Topical
3) By Application: Oncology, Genetic Disorders, Cardiovascular Diseases, Infectious Diseases, Neurological Disorders, Other Applications
4) By End-User: Hospitals, Clinical Research Organizations (CROs), Academic And Research Institutes, Pharmaceutical And Biotechnology Companies, Other End-Users
Subsegments:
1) By Adenoviral Vectors: Human Adenovirus Serotype Five, Human Adenovirus Serotype Two, Canine Adenovirus
2) By Adeno-Associated Viral Vectors: Adeno-Associated Virus Serotype One, Adeno-Associated Virus Serotype Two, Adeno-Associated Virus Serotype Five, Adeno-Associated Virus Serotype Eight, Other Adeno-Associated Viruses
3) By Lentiviral Vectors: Human Immunodeficiency Virus One, Human Immunodeficiency Virus Two
4) By Retroviral Vectors: Gammaretrovirus, Alpharetrovirus, Spumaretrovirus, Other Retroviruses
What Trends Are Shaping The Future Of The Viral Vector-Based Gene Therapy Market?
Leading firms within the viral vector-based gene therapy market are concentrating efforts on creating sophisticated platforms, like integrated manufacturing systems, with the goal of improving efficiency, strengthening dependability, and cutting down both the expense and duration of therapy manufacturing. These integrated manufacturing platforms represent holistic systems designed to simplify the complete gene therapy production journey, encompassing everything from vector conceptualization to the finished product, by employing standardized and highly productive methods. As an example, in October 2024, the US biotechnology company Forge Biologics Holdings LLC introduced its FUEL AAV Manufacturing Platform. This platform is a proprietary, suspension-based manufacturing system engineered for superior productivity and scalability. It incorporates a comprehensively integrated plasmid system and refined processes, which facilitates the strong and uniform creation of adeno-associated viral (AAV) vectors crucial for gene therapies, consequently expediting developer schedules from the research phase through to clinical trials.
Who Are The Established Players Within The Viral Vector-Based Gene Therapy Market?
Major companies operating in the viral vector-based gene therapy market are F. Hoffmann-La Roche AG, Novartis AG, BioMarin Pharmaceutical Inc., Sarepta Therapeutics Inc., Ultragenyx Inc., Rocket Pharmaceuticals Inc., PTC Therapeutics Inc., 4D Molecular Therapeutics Inc., Editas Medicine Inc., Regenxbio Inc., Oxford Biomedica plc, MeiraGTx Holdings plc, Voyager Therapeutics Inc., Solid Biosciences Inc., Sangamo Therapeutics Inc., Adverum Biotechnologies Inc., Passage Bio Inc., Dyno Therapeutics Inc., uniQure N.V., Bluebird Bio Inc.
Get The Full Viral Vector-Based Gene Therapy Market Report:
Which Region Is The Top Contributor To The Viral Vector-Based Gene Therapy Market By Share?
North America was the largest region in the viral vector-based gene therapy market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the viral vector-based gene therapy market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Viral Vector-Based Gene Therapy Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Viral Vector-Based Gene Therapy Market 2026, By The Business Research Company
Genes Therapy Market Report 2026
https://www.thebusinessresearchcompany.com/report/genes-therapy-global-market-report
Antiviral Combination Therapy Market Report
https://www.thebusinessresearchcompany.com/report/antiviral-combination-therapy-global-market-report
Oral Expectorant Market Report
https://www.thebusinessresearchcompany.com/report/oral-expectorant-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model