Pharmacovigilance And Drug Safety Software Market Outlook Highlighting Key Drivers And Market Evolution
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
By 2030, What Market Size Is The Pharmacovigilance And Drug Safety Software Market Expected To Reach Based On Its 2026 Value?
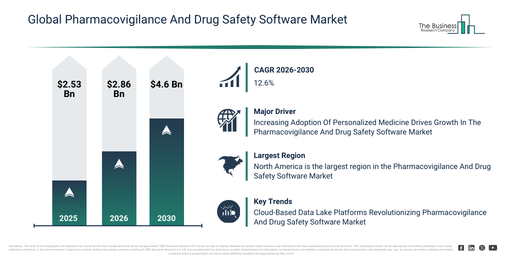
The pharmacovigilance and drug safety software market has seen substantial growth in recent years. Its value is expected to rise from $2.53 billion in 2025 to $2.86 billion in 2026, achieving a compound annual growth rate (CAGR) of 12.8%. This historical expansion can be attributed to an increase in adverse drug reaction reporting, the globalization of clinical trials, the implementation of more stringent drug safety regulations, an increase in pharmaceutical production, and the growing demand for post-marketing surveillance.
The pharmacovigilance and drug safety software market size is projected for substantial expansion over the upcoming years, reaching $4.6 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 12.6%. This anticipated growth during the forecast period is driven by factors such as the increasing deployment of AI-driven signal detection, the proliferation of biologics and specialty drugs, heightened regulatory scrutiny, a rise in the outsourcing of pharmacovigilance services, and the need for real-time safety analytics. Key developments expected in this period encompass the wider embrace of cloud-based pharmacovigilance platforms, a greater emphasis on monitoring real-world evidence, increased automation in reporting adverse events, the broadening of integrated drug safety systems, and a heightened requirement for regulatory compliance solutions.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=19140&type=smp
What Primary Drivers Are Shaping The Pharmacovigilance And Drug Safety Software Market?
The increasing need for personalized medicine is projected to boost the expansion of the pharmacovigilance and drug safety software market. Personalized medicine, also known as precision medicine, represents a healthcare approach that tailors treatment decisions to an individual’s genetic, biological, and lifestyle characteristics. This demand is escalating due to improved treatment outcomes, enhanced prevention and early detection strategies, and greater cost-effectiveness. The pharmacovigilance and drug safety software market supports personalized medicine by integrating pharmacogenetics, real-time data analytics, and post-marketing surveillance to optimize patient safety, monitor individualized treatments, and strengthen risk-management practices. For instance, in February 2024, according to the Personalized Medicine Coalition (PMC), a US-based non-profit organization, the FDA approved 16 novel personalized therapies for patients with rare diseases in 2023, compared to 6 approvals in 2022. Consequently, the growing demand for personalized medicine is driving the growth of the pharmacovigilance and drug safety software market.
What Are The Main Segments Within The Pharmacovigilance And Drug Safety Software Market Segment Structure?
The pharmacovigilance and drug safety software market covered in this report is segmented –
1) By Software Type: Adverse Event Reporting Software, Drug Safety Audits Software, Issue Tracking Software, Fully Integrated Software
2) By Delivery Mode: On-premise, Cloud-based
3) By End User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Business Process Outsourcing Firms, Other End Users
Subsegments:
1) By Adverse Event Reporting Software: Spontaneous Reporting Systems, EHR Integration Solutions, Mobile Reporting Applications
2) By Drug Safety Audits Software: Audit Management Solutions, Compliance Tracking Tools, Reporting And Analytics Modules
3) By Issue Tracking Software: Incident Reporting Systems, Workflow Management Tools, Collaboration Platforms
4) By Fully Integrated Software: Pharmacovigilance Platforms, Regulatory Compliance Systems, Data Analytics And Visualization Tools
What Market Trends Are Affecting The Pharmacovigilance And Drug Safety Software Market?
Companies operating in the **pharmacovigilance and drug safety software market** are concentrating on developing innovative technologies, such as cloud-based data lake platforms, to strengthen adverse event monitoring, improve regulatory compliance, and enhance overall patient safety across clinical trials and post-market surveillance activities. A cloud-based data lake platform refers to a scalable, centralized system capable of storing and analyzing large volumes of structured and unstructured safety data in real time, thereby enabling advanced analytics, AI/ML modeling, and streamlined case processing within pharmacovigilance workflows. For instance, in **December 2023**, **Thermo Fisher Scientific Inc., a US-based biotechnology company**, introduced CorEvidence, a proprietary cloud-based data lake platform engineered to optimize pharmacovigilance processes by improving safety data management and case processing efficiency, particularly supporting post-authorization safety studies within clinical research registries.
Who Are The Companies Participating In The Pharmacovigilance And Drug Safety Software Market?
Major companies operating in the pharmacovigilance and drug safety software market are IQVIA, Oracle Corporation, ArisGlobal, RXLogix, Anju Software Inc., EXTEDO GmbH, Sarjen Systems Pvt Ltd, Ennov Group, AB Cube, Clinevo Technologies, Accenture plc, International Business Machines Corporation, Capgemini SE, Cognizant Technology Solutions, Wipro Limited, Genpact, Indegene Limited, PharmaLex, Deloitte Touche Tohmatsu Limited, Parexel
Get The Full Pharmacovigilance And Drug Safety Software Market Report:
Where Is The Pharmacovigilance And Drug Safety Software Market Primarily Concentrated By Region?
North America was the largest region in the pharmacovigilance and drug safety software market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the pharmacovigilance and drug safety software market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Pharmacovigilance And Drug Safety Software Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Pharmacovigilance And Drug Safety Software Market 2026, By The Business Research Company
Pharmacovigilance Market Report 2026
https://www.thebusinessresearchcompany.com/report/pharmacovigilance-global-market-report
Patient Safety And Risk Management Software Market Report 2026
Therapeutic Drug Monitoring Market Report 2026
https://www.thebusinessresearchcompany.com/report/therapeutic-drug-monitoring-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at: marketing@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model