Pharmacovigilance Outsourcing Market Forecast With Insights On Demand And Industry Growth
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Changes In Market Value Are Expected For The Pharmacovigilance Outsourcing Market Over The 2026–2030 Period?
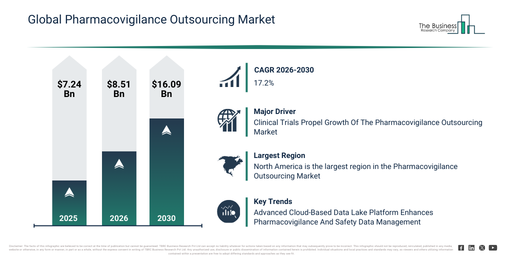
The pharmacovigilance outsourcing market has experienced rapid expansion in recent years. It is projected to grow from $7.24 billion in 2025 to $8.51 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 17.6%. The market’s historical growth can be attributed to an escalating number of new drug approvals, increased pharmacovigilance awareness among pharma companies, a deficiency in in-house pv expertise, intensifying regulatory scrutiny, and the broader expansion of global clinical trials.
The pharmacovigilance outsourcing market is projected to experience substantial expansion over the upcoming years. This market is anticipated to reach $16.09 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 17.2%. Key drivers for this growth during the forecast period include the incorporation of AI and data analytics into PV processes, a rise in outsourcing to contract research organizations (CROs) and business process outsourcing (BPO) providers, the embrace of cloud-based pharmacovigilance platforms, the widening scope of global safety reporting mandates, and increased investment in IT infrastructure and technological assistance. Significant trends predicted for the forecast period encompass a greater uptake of outsourced pharmacovigilance services by pharmaceutical companies, stricter regulatory compliance demands and reporting benchmarks, an increase in the automation of adverse drug reaction (ADR) capture and case handling, the development of knowledge management and data integration tools, and a heightened emphasis on risk management and quality assurance offerings.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=19651&type=smp
Which Key Factors Are Driving The Pharmacovigilance Outsourcing Market Growth?
The increasing volume of clinical trials is anticipated to boost the expansion of the pharmacovigilance outsourcing market. These trials are investigative studies performed to assess the safety, effectiveness, and possible adverse effects of novel therapies, medications, or medical instruments. The escalation in clinical trials stems from various factors, such as an increased emphasis on personalized medicine, the growing prevalence of chronic illnesses, favorable regulatory environments, and the global spread of research activities. Outsourcing pharmacovigilance during clinical trials improves drug safety oversight by utilizing specific knowledge and assets to guarantee adherence to regulatory mandates and enhance operational effectiveness. As an illustration, data from the Association of the British Pharmaceutical Industry (ABPI), a UK-based trade association, revealed that the number of industry clinical trials initiated in the UK rose to 426 in 2023, an increase from 411 in 2022. This particular finding was reported in December 2024. Consequently, the increased volume of clinical trials is fueling the expansion of the pharmacovigilance outsourcing market.
How Is The Pharmacovigilance Outsourcing Market Segmented Across Different Segment Categories?
The pharmacovigilance outsourcing market covered in this report is segmented –
1) By Type: Adverse Drug Reaction Capture (ADR), Case Processing, Reporting And Submission, Report Publishing, Quality Check, Risk Management, Knowledge Management, Enabling Architecture
2) By Service Provider: Contract Research Organization, Business Processing Outsourcing
3) By End User: Pharmaceutical Industry, Research Organization, Other End Users
Subsegments:
1) By Adverse Drug Reaction Capture (ADR): Automated ADR Reporting Tools, Manual ADR Reporting Solutions, ADR Data Mining Software
2) By Case Processing: Case Intake and Triage Services, Medical Review and Evaluation Services, Data Entry and Management Services
3) By Reporting And Submission: Regulatory Reporting Services, Submission to Health Authorities, Compliance Tracking Services
4) By Report Publishing: Clinical Study Report Publishing, Safety Update Report Publishing, Final Study Report Publishing
5) By Quality Check: Quality Assurance Services, Audit and Inspection Services, Compliance Monitoring Services
6) By Risk Management: Risk Assessment Services, Risk Minimization Strategies, Benefit-Risk Evaluation Services
7) By Knowledge Management: Data Management Solutions, Pharmacovigilance Database Management, Knowledge Sharing Platforms
8) By Enabling Architecture: IT Infrastructure Solutions, Data Integration Services, Technology Platform Support
Which Trends Are Contributing To Changes In The Pharmacovigilance Outsourcing Market?
Major companies within the pharmacovigilance outsourcing market are concentrating their efforts on creating advanced solutions, such as cloud-based data lake platforms. These innovations aim to optimize the handling of pharmacovigilance cases and the management of safety data, thereby improving clinical research registries and ensuring adherence to regulatory standards. A cloud-based data lake platform functions as a central storage facility, capable of holding and organizing vast quantities of raw data in its original form until it is needed. For example, in December 2023, Thermo Fisher Scientific Inc., a US-based biotechnology and laboratory equipment company, introduced CorEvidence. This platform was specifically designed to enhance clinical research registries and simplify pharmacovigilance case processing by integrating various data sources and refining the coding, classification, and reporting of adverse events. Its importance stems from its ability to offer scalable, compliant, and auditable safety management, bolstering CorEvitas clinical research registries, and aiding extended post-authorization safety studies across diverse therapeutic domains.
Which Organizations Play A Role In The Pharmacovigilance Outsourcing Market Landscape?
Major companies operating in the pharmacovigilance outsourcing market are ACCENTURE PLC, International Business Machines Corporation (IBM), Tata Consultancy Services, Capgemini SE, Cognizant Technology Solutions Corporation, IQVIA Holdings Inc., Wipro Limited, ICON PLC, Syneos Health, Genpact limited, Parexel International Corp, Veeva Systems Inc., Medpace Holdings Inc., ProPharma Group, Pharmalex GmbH, PrimeVigilance Ltd., Veristat LLC, United BioSource LLC, Lambda Therapeutic Research, ArisGlobal, Biomapas
Get The Full Pharmacovigilance Outsourcing Market Report:
https://www.thebusinessresearchcompany.com/report/pharmacovigilance-outsourcing-global-market-report
Which Region Is The Leading Market For The Pharmacovigilance Outsourcing Market?
North America was the largest region in the pharmacovigilance outsourcing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the pharmacovigilance outsourcing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Pharmacovigilance Outsourcing Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/pharmacovigilance-outsourcing-global-market-report
Browse Through More Reports Similar to the Global Pharmacovigilance Outsourcing Market 2026, By The Business Research Company
Pharmaceutical Drug Delivery Market Report 2026
https://www.thebusinessresearchcompany.com/report/pharmaceutical-drug-delivery-global-market-report
Veterinary Pharmaceuticals Market Report 2026
https://www.thebusinessresearchcompany.com/report/veterinary-pharmaceuticals-global-market-report
Pharmaceutical Sterility Testings Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at: marketing@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model
[KClientError] [REQ_ERR: OPERATION_TIMEDOUT] [KTrafficClient] Something is wrong.