Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Insights On Growth Drivers, Trends, And Opportunities
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Expansion In Market Value Is Forecasted For The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Between 2026 And 2030?
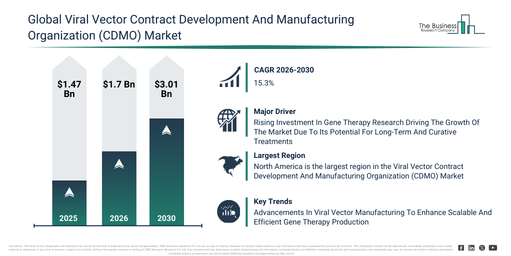
The viral vector contract development and manufacturing organization (CDMO) market size has shown swift growth in recent years. It is anticipated to increase from $1.47 billion in 2025 to $1.7 billion in 2026, achieving a compound annual growth rate (CAGR) of 15.6%. The historical expansion can be ascribed to a burgeoning clinical pipeline for gene therapies, the early adoption of outsourcing for viral vector production, an increase in academic research collaborations, the inherent limited in-house manufacturing capacities of biotech firms, and a rising number of regulatory approvals for viral vectors.
The viral vector contract development and manufacturing organization (cdmo) market is projected to experience substantial expansion over the coming years. This market is anticipated to reach $3.01 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 15.3%. Several factors contribute to this growth during the forecast period, including the increasing commercialization of gene and cell therapies, higher investments in specialized viral vector facilities, a rising need for scalable manufacturing solutions, the broadening of global cdmo collaborations, and advancements in high-yield vector production technologies. Key trends defining this period encompass an expansion of capacity for large-scale viral vector production, the increasing embrace of single-use manufacturing technologies, a growing requirement for comprehensive end-to-end cdmo services, a heightened emphasis on cGMP facilities that comply with regulations, and improved integration of advanced analytics in vector manufacturing.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=29238&type=smp
Which Primary Drivers Are Impacting The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Growth?
The increasing financial commitment to gene therapy research is projected to fuel the expansion of the viral vector contract development and manufacturing organization (CDMO) market in the future. Gene therapy investigations center on discovering techniques to modify or control genes within human cells, aiming to rectify genetic ailments or address diverse illnesses. Funding for gene therapy research is escalating because of its capacity to offer lasting or permanent treatments for genetic conditions that were previously incurable, thereby attracting significant attention from healthcare pioneers and financial backers alike. Viral vector CDMOs assist gene therapy studies by providing specialized expertise and cutting-edge manufacturing plants for producing high-quality viral vectors, essential for the secure and efficient delivery of therapeutic genes. As an illustration, in July 2025, the UK government, through the Office for Life Sciences, a governmental department, pledged up to $800 million (£600 million) towards establishing a leading, secure, and AI-ready health data platform globally. This platform will consolidate genomic, diagnostic, and clinical data across the population, converting NHS and wider healthcare data into a central point for international research studies and AI investments. Consequently, the increasing financial input into gene therapy research is propelling the expansion of the viral vector contract development and manufacturing organization (CDMO) market.
What Segment Categories Shape The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Segment Landscape?
The viral vector contract development and manufacturing organization (cdmo) market covered in this report is segmented –
1) By Service Type: Process Development, Manufacturing, Analytical Testing, Fill-Finish, Other Service Types
2) By Vector Type: Adenoviral Vectors, Lentiviral Vectors, Adeno-Associated Viral Vectors, Retroviral Vectors, Other Vector Types
3) By Workflow: Upstream Manufacturing, Downstream Manufacturing
4) By Application: Gene Therapy, Vaccines, Cell Therapy, Other Applications
5) By End-User: Pharmaceutical And Biotechnology Companies, Academic And Research Institutes, Other End-Users
Subsegments:
1) By Process Development: Upstream Process Development, Downstream Process Development, Analytical Development, Process Characterization
2) By Manufacturing: Clinical Manufacturing, Commercial Manufacturing, cGMP Manufacturing, Cell Bank Production, Fill-Finish Services
3) By Analytical Testing: Release Testing, Stability Testing, Bioassays, Characterization Studies, Regulatory Compliance Testing
4) By Fill-Finish: Aseptic Filling, Lyophilization, Final Packaging, Cold Chain Management, Labeling And Serialization
5) By Other Service Types: Regulatory Support, Supply Chain Management, Technology Transfer, Clinical Trial Support, Cold Chain Logistics
What Trends Are Transforming The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market?
Leading entities within the viral vector contract development and manufacturing organization (CDMO) market are prioritizing the creation of advanced solutions, such as AAV manufacturing solutions, to enable scalable and consistent production. AAV manufacturing solutions encompass methodologies and systems designed for the efficient production of safe and effective adeno-associated virus vectors vital for gene therapy. For instance, in August 2025, ProBio, a US-based contract development and manufacturing organization (CDMO), commenced cGMP Adeno-Associated Virus (AAV) manufacturing services at its modern 128,000 sq. ft. facility in Hopewell, New Jersey. This growth initiative seeks to fulfill the rising demand for high-quality viral vector production while showcasing ProBio’s commitment to fostering the advancement of innovative gene therapies. The Hopewell facility was purpose-built to offer comprehensive AAV manufacturing solutions in adherence to global regulatory and quality requirements. ProBio’s service portfolio now features fully integrated offerings like GMP plasmid DNA production, AAV vector manufacturing, and final drug product formulation with aseptic fill/finish, all housed in a single U.S. location. This streamlined strategy enhances coordination, reduces transitions between processes, and quickens timelines across the entire drug development lifecycle.
Who Are The Companies Operating Across The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Value Chain?
Major companies operating in the viral vector contract development and manufacturing organization (cdmo) market are Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, WuXi AppTec, Samsung Biologics Co. Ltd., FUJIFILM Biotechnologies, Oxford Biomedica Plc, Hillgene, Takara Bio Inc., SkyPharma Production SAS, GeneScript ProBio, Obio Technology Corp. Ltd., VectorBuilder, Charles River Laboratories Pvt. Ltd., Creative Biogene, Esco Aster Pte. Ltd., Genesail Biotech Co. Ltd., CEVEC Pharmaceuticals, Cell and Gene Therapy Catapult, CoJourney, Applied Biological Laboratories Inc.
Get The Full Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Report:
Which Region Holds The Highest Market Share In The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market?
North America was the largest region in the viral vector contract development and manufacturing organization (CDMO) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the viral vector contract development and manufacturing organization (cdmo) market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Viral Vector Contract Development And Manufacturing Organization (CDMO) Market 2026, By The Business Research Company
Viral Vector Manufacturing Market Report 2026
https://www.thebusinessresearchcompany.com/report/viral-vector-manufacturing-global-market-report
Viral And Non Viral Vector Manufacturing Market Report 2026
Oral Expectorant Market Report 2026
https://www.thebusinessresearchcompany.com/report/oral-expectorant-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at: marketing@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model