GMP Cell Therapy Consumables Market Trends Report 2025 – For Business Development and Strategy Teams
Claim your 30% discount on Global Market Reports with code ONLINE30. Limited time only.
What Is the Forecast for the GMP Cell Therapy Consumables Market Growth in 2025?
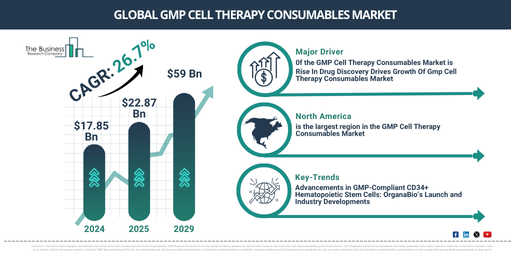
In recent times, a significant exponential growth has been observed in the market size of GMP cell therapy consumables. The market, which was worth $17.85 billion in 2024 is set to rise to $22.87 billion in 2025, reflecting a compound annual growth rate (CAGR) of 28.1%. This growth during the historical period is a result of increasing investments in regenerative medicine, progress in cell therapy research, clinically approved trials, higher prevalence of chronic diseases, and a burgeoning awareness about personalized medicine.
What Is the Forecast for the GMP Cell Therapy Consumables Market Size Through 2029?
In the coming years, the market size for GMP cell therapy consumables is projected to experience a steep rise. By 2029, it is predicted to expand to a massive $59 billion, with a compound annual growth rate (CAGR) of 26.7%. This prospective growth within the forecast period can be credited to factors such as the widening applications of cell therapy, a worldwide increase in cell therapy trials, regulatory backing for cell therapies, the advent of advanced cell therapy platforms, and the globalisation of cell therapy manufacturing. Key trends to look for within the forecast period encompass investments in manufacturing infrastructure, innovations in cell processing technology, collaborations and partnerships, digitization and data integration, as well as strategic alliances and mergers.
Download The Free Sample Report Here:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12014&type=smp
What are the Key Market Players in GMP Cell Therapy Consumables Market and How They’re Evolving?
Major companies operating in the GMP cell therapy consumables market include Thermo Fisher Scientific Inc., Fresenius Kabi AG, Danaher Corporation, Merck KGaA, Asahi Kasei Corporation, GE HealthCare Technologies Inc., Corning Incorporated, Avantor Inc., Lonza Group AG, Terumo Corporation, Catalent Inc., Sartorius AG, Bio-Techne Corp, Repligen Corporation, Miltenyi Biotec BV & Co KG, Genscript Biotech Corporation, Rentschler Biopharma SE, FUJIFILM Irvine Scientific Inc., BioLegend Inc., STEMCELL Technologies Inc., BioLife Solutions Inc., Abzena Ltd., Sino Biological Inc., ProBioGen AG, MaxCyte Inc., PromoCell GmbH, PeproTech Inc., Cellares Corp., Wilson Wolf Corporation, Cellexus Ltd.
What Are the Primary Growth Drivers in the GMP Cell Therapy Consumables Market?
The GMP cell therapy consumables market is anticipated to expand due to the increasing interest in drug discovery. This process, which aims to find new possible medicinal substances, involves a mix of computational, experimental, translational, and clinical modeling. Recent breakthroughs in areas such as molecular biology, genetics, bioinformatics, and high-throughput screening methodologies have drastically transformed the landscape of drug discovery. Such advancements allow researchers to gain a deeper understanding of disease processes, pinpoint potential drug targets, and more effectively sift through extensive compound libraries for viable drug candidates. This heightened activity in drug discovery fuels the expansion of the GMP cell therapy consumables market. As an illustration, the Food and Drug Administration, a US federal agency within the Department of Health and Human Services, reported in January 2023 that its Centre for Drug Evaluation and Research (CDER) had approved 37 new drugs and therapeutic biological products in 2022 for various uses. Thus, the acceleration in drug discovery efforts is directly influencing the growth of the GMP cell therapy consumables market.
Request For A Customized Report:
https://www.thebusinessresearchcompany.com/customise?id=12014&type=smp
What Are the Leading Segments in the Global GMP Cell Therapy Consumables Industry?
The GMP cell therapy consumables market covered in this report is segmented –
1) By Product: Kits, Reagents Or Molecular Biology reagents, Growth Factors Or Cytokines And Interleukins, Other Products
2) By Cell Therapy: NK Cell Therapy, Stem Cell Therapy, T-Cell Therapy, Other cell therapies
3) By Process: Cell Collection And Characterization Or Sorting And Separation, Cell Culture And Expansion Or Preparation, Cryopreservation, Cell Processing And Formulation, Cell Isolation And Activation, Cell Distribution Or Handling, Process Monitoring And Control Or Readministration Or Quality Assurance, Other Processes
4) By End-Use: Clinical, Commercial, Research
Subsegments:
1) By Kits: Cell Culture Kits, Cell Isolation Kits, Transfection Kits
2) By Reagents Or Molecular Biology Reagents: Buffers And Solutions, Enzymes, Staining Reagents
3) By Growth Factors Or Cytokines And Interleukins: Hematopoietic Growth Factors, Antibody Cytokines, Immunomodulatory Cytokines
3) By Other Products: Cell Culture Vessels, Bioreactors, Media And Supplements
What Are the Key Market Trends in the GMP Cell Therapy Consumables Industry?
Primary corporations in the GMP cell therapy consumables market are producing GMP-compliant CD34+ hematopoietic stem cells (HSCs) to aid advanced treatments. These GMP-compliant CD34+ HSCs are a unique stem cell type that display the CD34 marker and are created following Good Manufacturing Practice (GMP) protocols. For instance, OrganaBio, a pharmaceutical firm located in the United States, introduced HematoPAC-HSC-CB-GMP, an accessible resource of GMP-compliant CD34+ HSCs from fresh human cord blood in April 2024. OrganaBio uses its comprehensive knowledge in cell isolation and GMP production to generate high quantities of highly viable CD34+ HSCs within a day of collection. The goal of this launch is to aid in the creation of cutting-edge treatments, including those for blood cancers and genetic diseases.
Access The Full Report Here:
https://www.thebusinessresearchcompany.com/report/gmp-cell-therapy-consumables-global-market-report
What Is the Regional Outlook for the GMP Cell Therapy Consumables Market?
North America was the largest region in the GMP cell therapy consumables market in 2024. Asia-pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the GMP cell therapy consumables market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Purchase The Full Report Today:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=12014
This Report Delivers Insight On:
1. How big is the gmp cell therapy consumables market, and how is it changing globally?
2. Who are the major companies in the gmp cell therapy consumables market, and how are they performing?
3. What are the key opportunities and risks in the gmp cell therapy consumables market right now?
4. Which products or customer segments are growing the most in the gmp cell therapy consumables market?
5. What factors are helping or slowing down the growth of the gmp cell therapy consumables market?
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 3156230293
Asia +44 2071930708
Europe +44 2071930708
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model