Major Trends and Emerging Patterns in the Preclinical Assets Market: Charles River Laboratories And Valo Health Optimized Preclinical Assets
Claim your 30% discount on Global Market Reports with code ONLINE30. Limited time only.
What Are the Key Projections for the CAGR of the Preclinical Assets Market Size From 2025 to 2034?
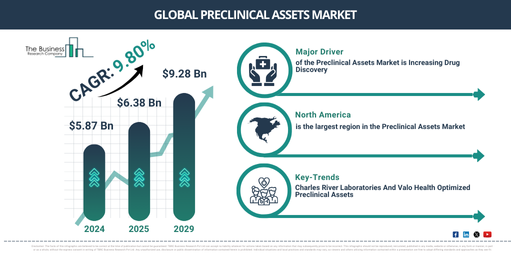
In recent times, there’s been considerable growth in the preclinical assets market size. The market, which is anticipated to increase from $5.87 billion in 2024 to $6.38 billion in 2025, is predicted to experience a compound annual growth rate (CAGR) of 8.8%. Factors contributing to the growth during the historic period include needs for drug development, advancements in biotechnology, regulatory requirements, growth in research funding, and collaborative research efforts.
The market size of preclinical assets is forecasted to experience a robust expansion in the upcoming years, reaching a value of$9.28 billion in 2029 with an annual growth rate (CAGR) of 9.8%. This projected growth over the forecast period can be linked to factors such as advances in precision medicine and personalized therapies, incorporation of artificial intelligence and machine learning, expansion of biobanking, increased attention to rare diseases, as well as governmental initiatives and financial support. Key emerging trends during the forecast period are expected to include the development of refined imaging techniques, the use of 3D organoid models, the application of multi-omics integration, improved predictive models, and the adoption of automation and high throughput screening.
Download a free sample to assess the report’s scope and structure:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12600&type=smp
What Are the Primary Drivers Supporting the Market Growth of the Preclinical Assets Market?
The surge in drug discovery is anticipated to fuel the expansion of the preclinical asset market. Drug discovery signifies the identification of chemical substances with potential for medicinal use, marking the inception of the creation of new candidate drugs. In this procedure, preclinical assets are critically important as they provide crucial data and proof that facilitates the choice of molecules for clinical development as potential candidates. For instance, the European Federation of Pharmaceutical Industries and Associations (EFPIA) revealed in June 2023 that the European pharmaceutical industry, based out of Belgium, saw significant production growth, escalating from $352.48 billion in 2021 to $369.95 billion in 2022. Therefore, the growing trend of drug discovery is propelling the expansion of the preclinical assets market.
Which Primary Segments of the Preclinical Assets Market Are Driving Growth and Industry Transformations?
The preclinical assetsmarket covered in this report is segmented –
1) By Service: Bioanalysis And DMPK (Drug Metabolism And Pharmacokinetic) Studies; Toxicology Testing; Compound Management; Safety Pharmacology; Other Services
2) By Model Type: Patient Derived Organoid (PDO) Model; Patient Derived Xenograft Model
3) By End User: Biopharmaceutical Companies; Government Institutes; Other End-Users
Subsegments:
1) By Bioanalysis And DMPK (Drug Metabolism And Pharmacokinetic) Studies: Pharmacokinetics (PK) Studies; Pharmacodynamics (PD) Studies; Bioanalytical Method Development
2) By Toxicology Testing: Acute Toxicity Testing; Chronic Toxicity Testing; Genotoxicity Testing; Carcinogenicity Testing
3) By Compound Management: Compound Library Management; Sample Storage And Retrieval; High-Throughput Screening Support
4) By Safety Pharmacology: Cardiovascular Safety Assessments; Neurotoxicity Evaluations; Respiratory Safety Evaluations
5) By Other Services: Regulatory Support; Preclinical Study Design And Consulting; Custom Assay Development
Request customized data on this market:
https://www.thebusinessresearchcompany.com/customise?id=12600&type=smp
Which Regions Are Key Players in the Growth of the #What Are the Primary Drivers Supporting the Market Growth of the Preclinical Assets Market?# Market?
North America was the largest region in the preclinical assets market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the preclinical assets market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa
What Are the Most Significant Market Trends in the Preclinical Assets Market?
The rise of product innovations is an increasing trend in the preclinical assets industry. Dominant companies in this sector are concentrating on delivering novel products and solutions to consolidate their position in the market and gain a competitive edge. In this regard, a development in April 2022 saw Charles River Laboratories International, an American pharmaceutical establishment with expertise in numerous preclinical and clinical laboratories, and Valo Health Inc., a US-based technology firm committed to reshaping drug discovery, introducing Logica. This AI-driven drug solution directly converts clients’ biological insights into enhanced preclinical assets. Logica takes advantage of Charles River’s exceptional preclinical understanding and Valo’s Opal Computational Platform-backed AI to offer clients revamped drug discovery via a cohesive, integrated solution that seamlessly translates goals to the nomination of candidates. Collaborating with Valo allows Charles River to merge its laboratory skills with molecular design technology, leading to an industry transformation by providing clients with highly researched leads and candidate while tying client expenses to value generation.
View the full report here:
https://www.thebusinessresearchcompany.com/report/preclinical-assets-global-market-report
What Parameters Are Used to Define the Preclinical Assets Market?
A preclinical asset refers to all clinical trial supplies or equipment for preclinical development that encompasses the activities that link drug discovery in the laboratory to the initiation of human clinical trials. This phase precedes human clinical trials and entails essential feasibility assessments, iterative evaluations, and safety data gathering, typically performed on laboratory animals.
Purchase the full report and get a swift delivery:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=12600
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model