Pyrogen Testing Market Outlook 2025: Mapping Growth, Innovation, and Regional Shifts
Claim your 30% discount on Global Market Reports with code ONLINE30. Limited time only.
How Fast Is The Pyrogen Testing Market Expected to Grow Between 2025 And 2029?
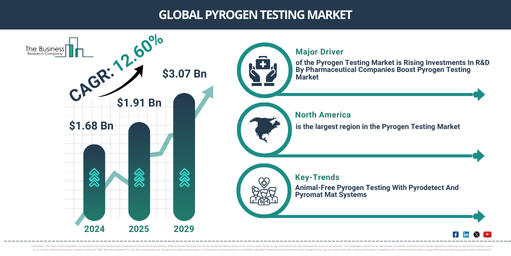
The market size for pyrogen testing has increased significantly over the last few years. It is forecasted to rise from $1.68 billion in 2024 to $1.91 billion in 2025 at a compound annual growth rate (CAGR) of 13.5%. Historical growth can be attributed to the expansion of the pharmaceutical and biotechnology industries, increasing regulations for drug safety, advancements in testing methodologies, a rise in the outbreaks of infectious diseases, and its adoption in the production of medical devices.
In the coming years, the pyrogen testing market size is set to experience substantial growth, with projections indicating a rise to$3.07 billion in 2029″, representing a compound annual growth rate (CAGR) of 12.6%. This growth in the predicted period can be attributed to several factors such as the development of custom pyrogen testing solutions, the rise of rapid and automated testing, advancements in in vitro pyrogen detection, the application of pyrogen testing in cellTherapy, and the implementation of eco-friendly and sustainable testing practices. Key trends during the forecast period also include point-of-care pyrogen testing, maintaining regulatory compliance in pyrogen testing, the use of AI for pyrogen detection and analysis, the provision of personalized and patient-specific pyrogen testing, and ensuring transparency and traceability in testing processes.
Download a free sample to assess the report’s scope and structure:
https://www.thebusinessresearchcompany.com/sample.aspx?id=4020&type=smp
What External and Internal Drivers Are Contributing to the Growth of thePyrogen Testing Market?
The escalating investment in R&D in the pharmaceutical and biotech sectors has fueled the growth of the pyrogen testing market. Pyrogen tests are utilized by pharmaceutical, biotech, and diverse drug production companies to assure the safe production and distribution of products free from infection. For example, in 2022, the innovative pharmaceutical sector, according to the Association of the British Pharmaceutical Industry, a UK-based commerce association, infused roughly £9 billion into UK R&D, potentially sprucing up the country’s health and economy. As such, the augmented research and investment from pharmaceutical companies propels the pyrogen testing market.
What Segment Types Define the Pyrogen Testing Market Structure?
The pyrogen testingmarket covered in this report is segmented –
1) By Test Type: LAL Test; In Vitro Test; Rabbit Test
2) By Product And Service: Assays, Kits, And Reagents; Instruments; Services
3) By Application: Pharmaceutical And Biologics; Medical Devices; Other Applications
Subsegments:
1) By Test Limulus Amebocyte Lysate (LAL) Test: Gel Clot LAL Test; Chromogenic LAL Test; Turbidimetric LAL Test
2) By In Vitro Test: Monocyte Activation Test (MAT); Recombinant Factor C (rFC) Assay; Other In Vitro Tests
3) By Rabbit Test: Pyrogen Test in Rabbits; Other Animal-Based Tests
Request customized data on this market:
https://www.thebusinessresearchcompany.com/customise?id=4020&type=smp
Which Geographic Areas Hold the Strongest Growth Potential in the Pyrogen Testing Market?
North America was the largest region in the pyrogen testing market in 2024. Asia-Pacific is expected to continue to be the fastest growing region in the pyrogen testing market share during the forecast period. The regions covered in the pyrogen testing market report include Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East and Africa.
What Long-Term Trends Are Transforming the Competitive Landscape of the Pyrogen Testing Market?
Firms are concentrating on the development of various non-animal pyrogen test procedures such as the monocyte activation test (MAT) to overcome the limitations of the rabbit pyrogen testing method and LAL test and to generate a human cure for animals. MAT offers a strong ability to predict pyrogenic activity, and it provokes fever response more effectively than the RPT or BET/LAL. The MAT pyrogen test method is employed to detect both endotoxin and non-endotoxin pyrogens, but the LAL test only detects endotoxin pyrogen. For example, Merck has centered its efforts on two MAT systems for pyrogen detection, PyroDetect and PyroMAT.
View the full report here:
https://www.thebusinessresearchcompany.com/report/pyrogen-testing-global-market-report
What Is the Definition of the Pyrogen Testing Market?
The pyrogen testing provide pyrogen testing to check the presence or absence of pyrogens in all aqueous parenteral. Pyrogen testing determines the presence of bacterial toxins in vaccines, biotechnological products, and drugs, which induces fever in humans. It also determines the presence of microbes and their metabolites in drugs during the manufacturing process.
Purchase the full report and get a swift delivery:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=4020
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model