Global Clinical Trial Site Management Organizations Market Report 2026–2035: Key Forecasts and Growth Areas
Uncover key drivers, emerging technologies, and competitive movements shaping the clinical trial site management organizations market from 2026–2035 with trusted insights from The Business Research Company
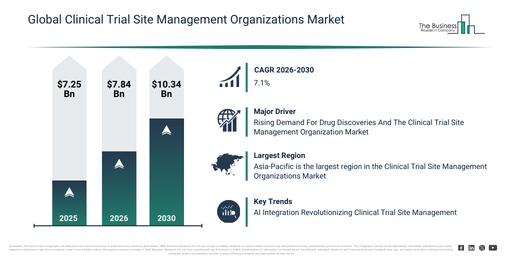
What level of growth is expected in the Clinical Trial Site Management Organizations Market between 2026 and 2030?
The market size for clinical trial site management organizations has experienced substantial growth in recent years. It is projected to expand from $7.25 billion in 2025 to $7.84 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 8.1%. Historically, this growth can be attributed to factors such as a greater number of multi-site clinical trials, an increased administrative burden on trial sites, the expansion of global trial networks, rising regulatory oversight at the site level, and the demand for enhanced site efficiency.
The clinical trial site management organizations market size is anticipated to experience robust growth in the upcoming years, projected to reach $10.34 billion by 2030, with a compound annual growth rate (CAGR) of 7.1%. This expansion during the forecast period is fueled by the increasing adoption of decentralized site operations, rising investments in tools for site digitization, a growing demand for faster site activation, the broadening of complex therapeutic trials, and an increasing emphasis on data quality and compliance. Prominent trends for this period include an increasing demand for specialized site management support, a rising focus on patient recruitment and retention services, the growing adoption of centralized site coordination models, the expansion of regulatory compliance support services, and enhanced utilization of digital site management tools.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=15766&type=smp
Which Drivers Are Expected To Impact The Clinical Trial Site Management Organizations Market During The Forecast Period?
The increasing requirement for drug discoveries is projected to fuel the expansion of the clinical trial site management organization market in the coming years. Drug discovery entails the identification and development of new medications or therapeutic compounds intended to treat diseases or improve health outcomes. The surge in drug discoveries is propelled by evolving medical necessities, including unmet diseases, emerging pathogens, drug resistance, and the aging population, all of which drive continuous innovation in pharmaceutical research and development. Clinical trial site management optimizes the coordination and oversight of clinical trials, thereby facilitating efficient drug discovery processes by ensuring compliance, effective data collection, and patient recruitment. For instance, in January 2024, according to Health Economists, a UK-based pharmaceutical and life science company, in 2023, there were 69 new active substances (NASs) approved globally, marking an increase from the previous year, including 24 first-in-class drugs approved in the U.S. This trend signifies a growing pipeline of innovative therapies. Therefore, the increasing demand for drug discoveries will drive the growth of the clinical trial site management organization market.
How Is The Clinical Trial Site Management Organizations Market Divided Into Segments?
The clinical trial site management organizations market covered in this report is segmented –
1) By Services: Site Management, Project Management, Regulatory, Onsite Monitoring
2) By Phase: Phase I, Phase II, Phase III, Phase IV
3) By Therapeutic Area: Oncology, Cardiology, Central Nervous System (CNS), Pain Management, Endocrine, Other Therapeutic Areas
Subsegments:
1) By Site Management: Site Selection And Feasibility, Patient Recruitment And Retention, Site Coordination And Administration
2) By Project Management: Project Planning And Scheduling, Budget And Resource Management, Risk Management And Compliance
3) By Regulatory: Regulatory Submissions And Approvals, Ethics And Institutional Review Board (IRB) Submissions, Regulatory Compliance And Audits
4) By Onsite Monitoring: Site Visits And Inspections, Data Quality Monitoring, Safety And Protocol Compliance Monitoring
Which Competitive Trends Are Impacting The Structure Of The Clinical Trial Site Management Organizations Market?
Leading companies in clinical trial site management organizations are increasingly integrating artificial intelligence into virtual trial platforms, focusing on clinical trial recruitment and engagement. This strategic move aims to optimize patient outreach, streamline data collection processes, and accelerate overall trial timelines. A clinical trial recruitment and engagement platform, through its AI integration, is designed to refine participant outreach, enhance the efficiency of recruitment processes, and improve patient engagement throughout the entire trial lifecycle. For instance, in October 2023, Elligo Health Research, a US-based healthcare-enabling research organization, joined forces with Avallano, a US-based clinical research platform provider, to introduce myTrialsConnect. This artificial intelligence (AI)-driven platform is dedicated to clinical trial recruitment and engagement, with the goal of optimizing participant enrollment and interaction in clinical trials. The platform allows patients to register as both trial participants and healthcare network patients, receiving alerts when they qualify for specific trials following automated medical record assessments and chatbot surveys.
Which Key Players Are Driving Competition In The Clinical Trial Site Management Organizations Market?
Major companies operating in the clinical trial site management organizations market are IQVIA Holdings Inc., ICON plc, PPD Inc., SGS SA, Syneos Health Inc., Charles River Laboratories International Inc., PAREXEL International Corporation, Medpace Holdings Inc, Premier Research Group Limited, WIRB-Copernicus Group, Worldwide Clinical Trials Inc., ClinChoice Inc., Clinigen Group plc, Precision for Medicine Inc., Synexus Clinical Research, Celerion Inc., Sarah Cannon Research Institute, Clinipace Worldwide Inc., Ergomed plc, Rho Federal Systems Division Inc., Veristat LLC, CTI Clinical Trial & Consulting Services, Clinedge, FOMAT Medical Research INC., Grand Pacific CRO, Xylem CRO, Access Clinical Research LLC, KV Consulting Corporation, Aurum Clinical Research, EthosExcel Inc.
Access The Complete Report For Deeper Market Insights:
How Does The Clinical Trial Site Management Organizations Market Perform Across Major Global Regions?
Asia-Pacific was the largest region in the clinical trial site management organizations market in 2025. The regions covered in the clinical trial site management organizations market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Clinical Trial Site Management Organizations Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=15766&type=smp
Browse Through More Reports Similar to the Global Clinical Trial Site Management Organizations Market 2026, By The Business Research Company
Clinical Trial Management System Ctms Market Report 2026
Clinical Trial Management Systems Market Report 2026
Clinical Trial Management System Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model