Which Emerging Trends Are Driving The Cell And Gene Therapy Clinical Trial Services Market Toward $6.02 Billion By 2029?
Claim your 20% discount on Global Market Reports with code ONLINE20. Limited time only.
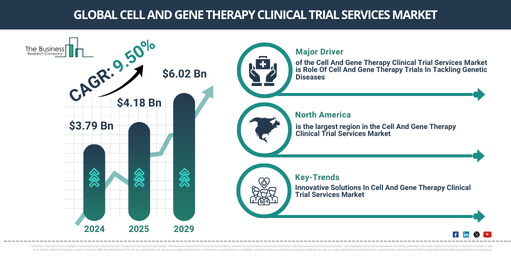
What CAGR And Valuation Are Anticipated For The Cell And Gene Therapy Clinical Trial Services Market?
The market size of clinical trial services for cell and gene therapy has seen a swift expansion in the past few years. The projections show that it would rise from $3.79 billion in 2024 to $4.18 billion in 2025, exhibiting a compound annual growth rate (CAGR) of 10.4%. Factors contributing to growth during the historical period include a surge in chronic disease cases, higher demand for efficacious treatments for rare genetic conditions, increased governmental support for regenerative medicine research, a deeper understanding of the molecular mechanisms of various illnesses, and a heightened awareness about the promise of personalized medicine.
It is anticipated that the market for cell and gene therapy clinical trial services will experience robust growth in the coming years, reaching a monetary value of $6.02 billion in 2029, expanding at a CAGR of 9.5%. The predicted growth over the forecasted period can be credited to the rising global instances of genetic and auto-immune disorders, increasing funding from both public and private sectors towards gene therapy research, the proliferation of innovative technologies, and an uptick in genetic mismatches. Other key trends projected through the forecast period encompass the utilization of AI, machine learning, personalized medicine, technological breakthroughs, and gene therapies.
Get A Free Sample Of The Report:
https://www.thebusinessresearchcompany.com/sample.aspx?id=15535&type=smp
What Core Drivers Are Expected To Influence The Cell And Gene Therapy Clinical Trial Services Market?
An upsurge in genetic disease occurrences is projected to stimulate the expansion of the cell and gene therapy clinical trial services market. Genetic diseases, conditions triggered by alterations to an individual’s DNA or genetic makeup, are on the rise for several reasons, including the better accessibility of genetic testing, the emergence of genetic mutations, and the widespread practice of marrying within the family lineage. These clinical trial services for cell and gene therapy tackle the growth in genetic diseases by promoting the creation of new treatments, customizing treatments to fit each patient’s needs, hastening the process of transforming research outcomes into actual clinical use, and widening the treatment choices for patients afflicted with genetic conditions. For instance, as per the data provided by the Cystic Fibrosis (CF) Foundation, a non-profit organization based in the U.S., in July 2022, there has been a jump in the count of cystic fibrosis patients. In 2022, cystic fibrosis impacted nearly 40,000 children and grown-ups in the U.S., and a total of over 105,000 people across the globe have been identified with the condition. Hence, the escalating trend of genetic diseases is propelling the cell and gene therapy clinical trial services market’s growth.
What Are The Main Segment Classifications In The Cell And Gene Therapy Clinical Trial Services Market?
The cell and gene therapy clinical trial servicesmarket covered in this report is segmented –
1) By Service: Clinical Trial Design And Planning; Supply And Logistic Services; Regulatory Affairs And Compliance; Data Management And Biostatics; Site Management And Monitoring; Other Services
2) By Therapy Type: Gene Therapy; Cell Therapy; Gene Modified Cell Therapy
3) By Indication: Oncology; Hematology; Metabolic Disorders; Infectious Diseases; Neurology; Cardiovascular Diseases; Musculoskeletal Disorders; Other Indications
4) By End-Use: Pharmaceutical And Biotechnology Companies; Contract Research Organizations; Academic And Research Institutes; Other End-Users
Subsegments:
1) By Clinical Trial Design And Planning: Protocol Development; Trial Feasibility Studies; Trial Design Consultation; Patient Recruitment And Enrollment Strategy; Risk-Based Monitoring Plans
2) By Supply And Logistic Services: Clinical Trial Supply Chain Management; Cold Chain Logistics; Packaging And Labeling Services; Transportation And Distribution Of Biological Samples; Customs And Import/Export Services
3) By Regulatory Affairs And Compliance: Regulatory Strategy And Consulting; Regulatory Submission And Documentation; Compliance With GMP (Good Manufacturing Practices); Regulatory Pathway Consultation; Clinical Trial Applications (CTAs)
4) By Data Management And Biostatistics: Clinical Data Management (CDM); Statistical Analysis And Reporting; Clinical Data Monitoring; Data Integration And Validation; Electronic Data Capture (EDC) Solutions
5) By Site Management And Monitoring: Site Selection And Initiation; Clinical Monitoring And Site Visits; Patient Recruitment And Retention; Site Training And Support; Site Performance Monitoring
6) By Other Services: Patient Advocacy And Engagement; Real-World Evidence (RWE) Studies; Post-Trial Services; Medical Writing And Documentation; Clinical Trial Auditing And Inspection
How Are Market Trends Influencing Competition In The Cell And Gene Therapy Clinical Trial Services Market?
Prominent businesses in the market for cell and gene therapy clinical trial services are increasingly investing in enhanced, customer relationship management system technology to boost efficiency levels and establish a competitive advantage within the market. The application of customer relationship management systems within cell and gene therapy clinical trial services can streamline the coordination and communication between trial participants, healthcare providers, and other stakeholders during the clinical trial process. To illustrate, in April 2023, AmerisourceBergen Corporation, a healthcare firm from the US, unveiled its Cell and Gene Therapy (CGT) Integration Hub. This platform, driven by their customer relationship management (CRM) system, is designed to enhance connectivity and smooth the processes across the cell and gene therapy treatment pathway. It aims to simplify care coordination by giving physicians and patient services teams improved oversight over therapy development and delivery, through features like quick benefits investigation, real-time status monitoring, and proactive alerts. The objective of CGT Integration Hub is to reduce obstacles and refine the overall experience of both patients and providers related to cell and gene therapies.
Which Companies Are Contributing Significantly To The Cell And Gene Therapy Clinical Trial Services Market Growth?
Major companies operating in the cell and gene therapy clinical trial services market are Thermo Fisher Scientific Inc., Sharp Services LLC, IQVIA, Laboratory Corporation of America Holdings, ICON plc, Syneos Health Inc., Catalent Pharma Solutions Inc., Charles River Laboratories International Inc., Parexel International Corporation, PRA Health Sciences Inc., Covance Inc., Medpace Holdings Inc., BioClinica Inc., Precision Medicine Group LLC, Worldwide Clinical Trials LLC, Clinigen Group plc, Evidera Inc., Advarra LLC, Veristat LLC, Clinipace Inc., Celonic AG, Cromsource Inc., Novotech Pty Ltd., MedSource Holdings Inc., Frontage Laboratories Inc.
Access The Complete Report Here:
Which Region Holds The Greatest Opportunity For Cell And Gene Therapy Clinical Trial Services Market Expansion?
North America was the largest region in the cell and gene therapy clinical trial services market in 2024. It is expected to be the fastest-growing region in the forecast period. The regions covered in the cell and gene therapy clinical trial services market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Customize Your Report Here:
https://www.thebusinessresearchcompany.com/customise?id=15535&type=smp
Browse Through More Reports Similar to the Global Cell And Gene Therapy Clinical Trial Services Market 2025, By The Business Research Company
Cell Therapy Technologies Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/cell-therapy-technologies-global-market-report
Cells Therapy Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/cells-therapy-global-market-report
Cell Surface Marker Detection Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/cell-surface-marker-detection-global-market-report
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model