Clinical-Stage Biotechnology Market Report 2026: Strategic Insights and Revenue Outlook
Uncover key drivers, emerging technologies, and competitive movements shaping the clinical-stage biotechnology market from 2026–2035 with trusted insights from The Business Research Company
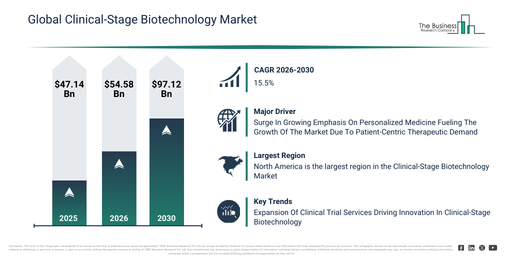
How much is the Clinical-Stage Biotechnology Market valued at in 2026, and what valuation is forecast for 2030?
The clinical-stage biotechnology market size has expanded significantly over recent years. It is anticipated to increase from $47.14 billion in 2025 to $54.58 billion in 2026, reflecting a compound annual growth rate (CAGR) of 15.8%. The historical growth trajectory is largely attributable to factors such as increased venture funding directed towards biotech startups, enhanced success rates observed in early-phase trials, the proliferation of academic-industry research collaborations, advancements in clinical trial infrastructure, and the escalating prevalence of complex diseases.
The clinical-stage biotechnology market is set to experience rapid expansion in the upcoming years. It is expected to grow to $97.12 billion by 2030, achieving a compound annual growth rate (CAGR) of 15.5%. This growth during the forecast period can be attributed to increasing investments in late-stage clinical programs, a rising demand for novel therapeutics, the expansion of precision medicine pipelines, the growing integration of digital trial management tools, and increasing regulatory support for innovative therapies. Key trends anticipated in the forecast period include the increasing advancement of pipeline assets into human trials, the rising adoption of adaptive clinical trial designs, the growing use of biomarkers in patient stratification, the expansion of multinational clinical studies, and an enhanced focus on translational research.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28173&type=smp
What Drivers Are Influencing The Growth Of The Clinical-Stage Biotechnology Market?
The expanding focus on personalized medicine is poised to accelerate the growth of the clinical-stage biotechnology market in the coming years. Personalized medicine signifies the practice of customizing medical treatment to suit the unique attributes of each patient, primarily by leveraging genetic, biomarker, and molecular data. This heightened emphasis on personalized medicine is growing because it empowers doctors to adapt cancer treatments to a patient’s genetic profile, leading to improved effectiveness and a reduction in unnecessary side effects. Clinical-stage biotechnology plays a crucial role in supporting personalized medicine through the development of targeted therapies that correspond with an individual’s genetic profile, thereby enhancing treatment effectiveness and safety. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based non-profit organization, the FDA sanctioned 16 new personalized therapies for patients with rare diseases in 2023, which was a notable increase from just six approvals recorded in 2022. Thus, the increasing importance of personalized medicine is anticipated to propel the expansion of the clinical-stage biotechnology market.
What Segments Are Included Within The Clinical-Stage Biotechnology Market?
The clinical-stage biotechnology market covered in this report is segmented –
1) By Product Type: Therapeutics, Diagnostics, Vaccines, Other Product Types
2) By Phase: Phase I, Phase II, Phase III
3) By Application: Oncology, Infectious Diseases, Cardiovascular Diseases, Neurology, Other Applications
4) By End-User: Hospitals, Research Institutes, Diagnostic Centers, Other End-Users
Subsegments:
1) By Therapeutics: Monoclonal Antibodies, Cell Therapy, Gene Therapy, Recombinant Proteins, Peptide Therapeutics, Small Molecule Therapeutics
2) By Diagnostics: Molecular Diagnostics, Immunoassays, Genetic Testing, Biomarker Testing, Point Of Care Diagnostics
3) By Vaccines: Live Attenuated Vaccines, Inactivated Vaccines, Subunit Vaccines, mRNA Vaccines, Viral Vector Vaccines
4) By Other Product Types: Regenerative Medicine Products, Research Reagents, Companion Diagnostics, Medical Devices
Which Trends Are Guiding The Evolution Of The Clinical-Stage Biotechnology Market?
Leading companies within the clinical-stage biotechnology market are prioritizing the creation of novel solutions, including clinical trial services, to expedite drug development, boost patient recruitment and retention, and strengthen adherence to regulations. These services encompass expert assistance and strategies for designing, overseeing, and executing clinical studies, aiming to assess the safety and efficacy of novel drugs or treatments. As an illustration, in November 2023, Ichor Life Sciences, Inc., a US-based biotechnology firm, introduced a new division named Ichor Clinical Trial Services. This new service broadens their existing contract research organization (CRO) capabilities, providing comprehensive support to biotechnology and pharmaceutical clients across the full spectrum of drug development, ranging from initial preclinical research to advanced clinical trials and eventual FDA approval. Ichor Clinical offers tailored solutions, covering aspects such as protocol development, identifying and qualifying study sites, devising recruitment and retention plans, and providing complete CRO assistance. The division’s objective is to tackle distinct client difficulties and intricate situations, for instance, aiding ongoing trials facing enrollment or operational hurdles, employing a customized and flexible methodology.
Who Are The Primary Competitors In The Global Clinical-Stage Biotechnology Market?
Major companies operating in the clinical-stage biotechnology market are Sarepta Therapeutics Inc., BioAge Labs Inc., PTC Therapeutics Inc., Evotec SE, Ultragenyx Pharmaceutical Inc., Rocket Pharmaceuticals Inc., Ventyx Biosciences Inc., Sangamo Therapeutics Inc., REGENXBIO Inc., Galapagos NV, Arvinas Inc., Editas Medicine Inc., Zymeworks Inc., CureVac N.V., Precision BioSciences Inc., 4D Molecular Therapeutics Inc., Domain Therapeutics S.A., ProQR Therapeutics N.V., Tonix Pharmaceuticals Holding Corp., Gilgamesh Pharmaceuticals Inc., T-Cure Bioscience Inc., Opna Bio Inc., EXUMA Biotech Inc., Prime Medicine Inc.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/clinical-stage-biotechnology-global-market-report
Which Global Regions Are Shaping The Competitive Landscape Of The Clinical-Stage Biotechnology Market?
North America was the largest region in the clinical-stage biotechnology market in 2025. The regions covered in the clinical-stage biotechnology market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Clinical-Stage Biotechnology Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=28173&type=smp
Browse Through More Reports Similar to the Global Clinical-Stage Biotechnology Market 2026, By The Business Research Company
Biotechnology Services Market Report 2026
https://www.thebusinessresearchcompany.com/report/biotechnology-services-global-market-report
Animal Biotechnology Market Report 2026
https://www.thebusinessresearchcompany.com/report/animal-biotechnology-global-market-report
Synthetic Biology Market Report 2026
https://www.thebusinessresearchcompany.com/report/synthetic-biology-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model
[KClientError] [REQ_ERR: OPERATION_TIMEDOUT] [KTrafficClient] Something is wrong.