Global Clinical Trial Biorepository And Archiving Solutions Industry Trends and Market Forecast 2026–2030
Uncover key drivers, emerging technologies, and competitive movements shaping the clinical trial biorepository and archiving solutions market from 2026–2035 with trusted insights from The Business Research Company
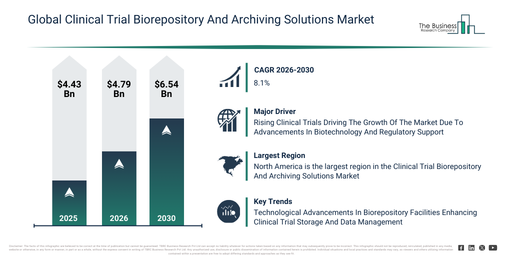
By how much is the Clinical Trial Biorepository And Archiving Solutions Market expected to grow between 2026 and 2030?
The clinical trial biorepository and archiving solutions market has shown strong growth in recent years. It is anticipated to expand from $4.43 billion in 2025 to $4.79 billion in 2026, achieving a compound annual growth rate (CAGR) of 8.1%. This market’s past growth can be ascribed to a rise in clinical trial volumes, more demanding regulatory documentation requirements, the broadening of pharmaceutical research and development activities, an increase in biobanking practices, and the globalization of clinical research.
The clinical trial biorepository and archiving solutions market is projected to demonstrate significant expansion in the upcoming years. It is expected to attain a size of $6.54 billion by 2030, progressing at a compound annual growth rate (CAGR) of 8.1%. This anticipated growth during the forecast period is attributable to several factors, including the surge in precision medicine trials, the expanding biopharmaceutical pipelines, an increasing requirement for long-term sample retention, the wider adoption of decentralized clinical trials, and more rigorous regulatory audit demands. Prominent trends observed within the forecast timeframe encompass an escalating demand for long-term biospecimen storage, the rising criticality of regulatory-compliant archiving practices, the global proliferation of clinical trial activities, an increased utilization of centralized biorepositories, and a concentrated focus on ensuring data integrity and sample traceability.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=23341&type=smp
Which Drivers Are Influencing Market Acceleration In The Clinical Trial Biorepository And Archiving Solutions Market?
A surge in clinical trials is projected to fuel the expansion of the clinical trial biorepository and archiving solutions market moving forward. These trials are research endeavors performed on human subjects to assess the safety, efficacy, and adverse effects of novel medical therapies or pharmaceuticals. The rise in clinical trial numbers stems from progress in medical research and the broadening scope of personalized medicine, demanding more precise and varied clinical investigations. Clinical trial biorepository and archiving solutions facilitate the growing volume of clinical trials by guaranteeing secure long-term storage, straightforward retrieval of biological specimens, and adherence to regulatory requirements, thereby improving data dependability and speeding up subsequent research. To illustrate this trend, in December 2024, the Association of the British Pharmaceutical Industry (ABPI), a UK-based association, reported an increase in industry-led clinical trial activity in the UK for the second year running, escalating from 411 trials in 2022 to 426 in 2023. Consequently, the expanding volume of clinical trials is a key driver for the growth of the clinical trial biorepository and archiving solutions market.
What Leading Segments Are Studied In The Clinical Trial Biorepository And Archiving Solutions Market?
The clinical trial biorepository and archiving solutions market covered in this report is segmented –
1) By Product: Preclinical Products, Clinical Products
2) By Service: Biorepository Services, Archiving Solution Services
3) By Application: Research And Development, Clinical Trials, Regulatory Compliance, Drug Development
4) By End User: Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations, Academic Institutions
Subsegments:
1) By Preclinical Products: Biospecimen Storage, Sample Processing And Preservation, Inventory Management Solutions, Data Management And Informatics
2) By Clinical Products: Biological Sample Archiving, Drug Compound Storage, Regulatory Compliance Solutions, Temperature-Controlled Storage
What Trends Are Projected To Support The Growth Of The Clinical Trial Biorepository And Archiving Solutions Market?
Leading companies active in the clinical trial biorepository and archiving solutions market are concentrating on developing innovative solutions, such as advanced biorepository facilities, to enhance storage, data management, and compliance within clinical research. A state-of-the-art biorepository facility signifies a sophisticated storage and management hub equipped with cutting-edge technology for preserving biological samples and clinical trial data. For example, in January 2025, Azenta Life Sciences, a US-based biotech company, launched a state-of-the-art biorepository facility in Greater Boston to support the biotech and life sciences industries with advanced sample storage and management. The facility facilitates the efficient handling of research, clinical trials, and biobanking, concurrently offering secure, compliant offsite storage solutions for clinical trials.
Which Key Market Players Are Investing In Expansion And Innovation Within The Clinical Trial Biorepository And Archiving Solutions Market?
Major companies operating in the clinical trial biorepository and archiving solutions market are Thermo Fisher Scientific Inc., GE HealthCare, Labcorp Holdings Inc., IQVIA, ICON plc, Avantor Inc., VWR International LLC, Charles River Laboratories, Parexel, Clinigen Limited, Inotiv Inc., Medpace Holdings Inc., LabConnect LLC, Azenta Life Sciences, HSRL Holdings LLC, EPL Archives, Manuh Solutions LLC, EMINENT Services Corporation, Boca Biolistics LLC, Astoriom
Access The Complete Report For Deeper Market Insights:
Which Regions Are Poised For Strategic Growth In The Clinical Trial Biorepository And Archiving Solutions Market?
North America was the largest region in the clinical trial biorepository and archiving solutions market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the clinical trial biorepository and archiving solutions market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Clinical Trial Biorepository And Archiving Solutions Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=23341&type=smp
Browse Through More Reports Similar to the Global Clinical Trial Biorepository And Archiving Solutions Market 2026, By The Business Research Company
Clinical Trial Supplies Market Report 2026
https://www.thebusinessresearchcompany.com/report/clinical-trial-supplies-global-market-report
Clinical Trial Management Systems Market Report 2026
Biological Sample Collection Kits Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model