Clinical Trial Software Market (2026–2030): Expansion Trends
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
How Large Will The Clinical Trial Software Market Be By 2030 Compared To Its 2026 Market Size?
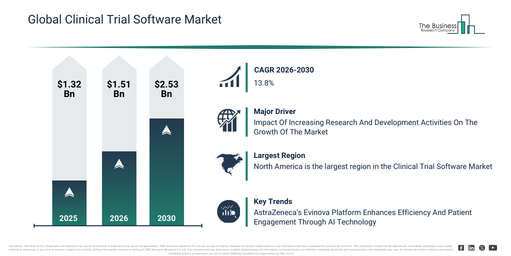
The clinical trial software market has seen rapid expansion in recent years. Its valuation is set to climb from $1.32 billion in 2025 to $1.51 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 14.1%. The market’s growth in the historical period was primarily influenced by the increasing digitization of clinical trial processes, the rising volume of clinical trial data, a growing necessity for real-time trial visibility, the expansion of global clinical research activities, and the availability of advanced software platforms.
The clinical trial software market is anticipated to experience substantial growth in the upcoming years. It is projected to expand to $2.53 billion by 2030, achieving a compound annual growth rate (CAGR) of 13.8%. This growth during the forecast period can be attributed to the increasing adoption of virtual and decentralized trials, rising investments in SaaS-based trial solutions, the growing integration of artificial intelligence in trial workflows, the expansion of remote patient monitoring, and an increased regulatory focus on data integrity. Major trends expected in this period include the rising adoption of cloud-based clinical trial software, increased use of AI-powered data analytics tools, growing demand for decentralized trial software platforms, the expansion of integrated EDC and CTMS solutions, and an enhanced focus on data security and compliance.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/report/clinical-trial-software-global-market-report
What Leading Drivers Are Supporting The Clinical Trial Software Market Expansion?
Elevated engagement in research and development is anticipated to boost expansion within the clinical trial software market. These activities involve a structured methodology for exploring, creating, and evaluating novel products or technologies, aiming to enhance current solutions or develop completely new ones. The surge in R&D efforts stems from a rising industry-wide need for innovation, driven by technological progress, intense market competition, and the necessity for sustainable options, prompting organizations to allocate greater resources toward crafting new products, processes, and services. Clinical trial software supports research and development by optimizing the planning, implementation, and oversight of clinical trials, essential for validating new medications and treatments. As an illustration, in 2023, the UK government’s net expenditure on research and development (R&D) reached £17.4 ($22.1 billion) billion, an increase from £16.1 billion ($20.45 billion) in 2022, signifying an 8.2% rise, according to the Office for National Statistics, a UK-based government agency. Consequently, the escalating engagement in research and development acts as a catalyst for the expansion of the clinical trial software market.
Which Segment Categories Are Included In The Clinical Trial Software Market Segment Analysis?
The clinical trial software market covered in this report is segmented –
1) By Deployment: On-Premises, Web-Based Clinical Trial Software, Cloud-Based Clinical Trial Software, Other Deployments
2) By Software: Electronic Data Capture (EDC), Electronic Clinical Outcome Assessment (eCOA) Or Electronic Patient-Reported Outcome (ePRO), Electronic Informed Consent
3) By End-User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations (CROs), Medical Device Manufacturers, Other End-Users
Subsegments:
1) By On-Premises: Enterprise Clinical Trial Management Systems, Site-Based Clinical Trial Solutions
2) By Web-Based Clinical Trial Software: Electronic Data Capture (EDC) Systems, Clinical Trial Management Systems (CTMS), Randomization And Trial Supply Management (RTSM)
3) By Cloud-Based Clinical Trial Software: Software-as-a-Service (SaaS) Platforms, AI-Powered Clinical Trial Solutions, Remote Monitoring And Decentralized Trial Systems
4) By Other Deployments: Hybrid Clinical Trial Solutions, Custom-Built Clinical Trial Software
What Emerging Trends Are Seen In The Clinical Trial Software Market?
Leading companies within the clinical trial software market are prioritizing the creation of advanced technologies, including unified trial solutions, aiming to improve data gathering and integration, boost patient involvement, streamline trial procedures, decrease expenses, and accelerate the entire drug development timeline. A Unified Trial Solution is defined as a comprehensive digital environment designed to optimize and elevate the clinical trial process. As an illustration, in November 2023, AstraZeneca plc, a pharmaceutical firm based in the UK, launched Evinova to facilitate the use of AI in clinical research. This innovative platform incorporates distinct functionalities, such as automated patient recruitment, which utilizes AI algorithms to rapidly match candidates with trial criteria, thereby significantly cutting down enrollment periods. It additionally integrates predictive modeling tools that examine historical and current data to anticipate patient reactions, thereby improving the structure and effectiveness of trials. Its intuitive interface promotes enhanced participant involvement by offering digital channels for interaction and feedback, making it simpler for participants to convey their experiences.
Who Are The Active Companies Shaping The Clinical Trial Software Market?
Major companies operating in the clinical trial software market are Oracle Corporation, Veeva Systems, Medidata Solutions, Signant Health Inc., Clario Inc., Greenphire Inc., Medrio Inc., Arisglobal LLC, Anju Software Inc., ClinCapture Inc., OpenClinica LLC, Castor, Viedoc, Clinion, OmniComm (TrialMaster), Curebase, Clinevo, RealTime-CTMS, REDCap Cloud, Zelta, Trialytix, Florence eBinders, Dot Compliance, ClinPlus CTMS, nQuery, Jeeva Clinical Trials
Get The Full Clinical Trial Software Market Report:
https://www.thebusinessresearchcompany.com/report/clinical-trial-software-global-market-report
Which Region Leads The Clinical Trial Software Market In Overall Market Size?
North America was the largest region in the clinical trial software market in 2025. Europe is expected to be the fastest-growing region in the forecast period. The regions covered in the clinical trial software market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Clinical Trial Software Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/clinical-trial-software-global-market-report
Browse Through More Reports Similar to the Global Clinical Trial Software Market 2026, By The Business Research Company
Clinical Trial Supplies Market Report 2026
https://www.thebusinessresearchcompany.com/report/clinical-trial-supplies-global-market-report
Clinical Trial Management System Market Report 2026
Virtual Clinical Trials Market Report 2026
https://www.thebusinessresearchcompany.com/report/virtual-clinical-trials-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model