Global Clinical Trial SMO Market (2026–2030): Strategic Insights
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Is The Projected Increase In Market Value Of The Clinical Trial Site Management Organizations Market During 2026–2030?
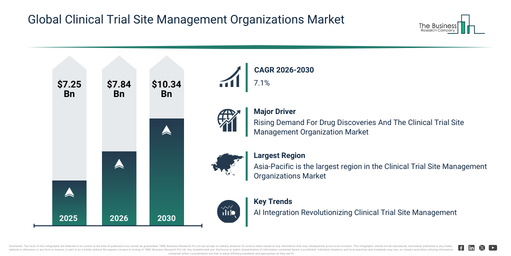
The clinical trial site management organizations market size has seen robust growth in recent years. It is forecast to rise from $7.25 billion in 2025 to $7.84 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 8.1%. This historical expansion can be attributed to factors such as an increasing number of multi-site clinical trials, a growing administrative burden on trial sites, the expansion of global trial networks, heightened regulatory oversight at the site level, and the demand for improved site efficiency.
The clinical trial site management organizations market size is anticipated to undergo significant expansion in the coming years. It is projected to reach $10.34 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 7.1%. This expected growth during the forecast period stems from factors such as the increasing embrace of decentralized site operations, heightened investments in site digitization tools, a rising need for quicker site activation, the expansion of complex therapeutic trials, and a greater emphasis on data quality and compliance. Noteworthy trends in the forecast period include an escalating demand for specialized site management support, a growing focus on patient recruitment and retention services, the increasing adoption of centralized site coordination models, an expansion of regulatory compliance support services, and the enhanced application of digital site management tools.
Access Your Free Sample Report For In-Depth Market Analysis:
Which Key Factors Are Driving The Clinical Trial Site Management Organizations Market Growth?
The increasing need for novel drug discoveries is anticipated to boost the expansion of the clinical trial site management organization market in the future. Drug discovery involves the process of identifying and developing new medications or therapeutic agents to address illnesses or enhance health outcomes. The surge in drug discovery efforts is driven by evolving medical requirements, such as previously unmet diseases, novel pathogens, antibiotic resistance, and an aging global demographic, which collectively stimulate ongoing innovation in pharmaceutical research and development. Clinical trial site management streamlines the coordination and supervision of clinical trials, thereby enabling more efficient drug discovery by guaranteeing regulatory compliance, accurate data gathering, and successful patient enrollment. As an illustration, data from Health Economists, a UK-based pharmaceutical and life science company, indicates that in January 2024, a total of 69 new active substances (NASs) were approved worldwide in 2023, marking an increase from the prior year. This figure included 24 first-in-class drugs that received approval in the U.S., highlighting an expanding portfolio of groundbreaking treatments. Consequently, the growing requirement for drug discoveries will stimulate the expansion of the clinical trial site management organization market.
How Is The Clinical Trial Site Management Organizations Market Broken Down By Segment Categories?
The clinical trial site management organizations market covered in this report is segmented –
1) By Services: Site Management, Project Management, Regulatory, Onsite Monitoring
2) By Phase: Phase I, Phase II, Phase III, Phase IV
3) By Therapeutic Area: Oncology, Cardiology, Central Nervous System (CNS), Pain Management, Endocrine, Other Therapeutic Areas
Subsegments:
1) By Site Management: Site Selection And Feasibility, Patient Recruitment And Retention, Site Coordination And Administration
2) By Project Management: Project Planning And Scheduling, Budget And Resource Management, Risk Management And Compliance
3) By Regulatory: Regulatory Submissions And Approvals, Ethics And Institutional Review Board (IRB) Submissions, Regulatory Compliance And Audits
4) By Onsite Monitoring: Site Visits And Inspections, Data Quality Monitoring, Safety And Protocol Compliance Monitoring
Which Trends Are Guiding The Direction Of The Clinical Trial Site Management Organizations Market?
Leading companies in the clinical trial site management sector are integrating artificial intelligence into virtual trial platforms, focusing on clinical trial recruitment and engagement. This strategic move aims to optimize patient outreach efforts, streamline data collection processes, and accelerate overall trial timelines. Specifically, a clinical trial recruitment and engagement platform leveraging AI is designed to enhance participant outreach, simplify recruitment procedures, and improve patient engagement throughout the entire trial lifecycle. As an illustration, in October 2023, Elligo Health Research, a US-based healthcare research organization, partnered with Avallano, a US-based clinical research platform provider, to unveil myTrialsConnect. This AI-powered clinical trial recruitment and engagement platform is engineered to optimize participant enrollment and interaction in clinical trials. It enables patients to register as trial participants and healthcare network patients, subsequently receiving notifications when they qualify for specific trials based on automated medical record assessments and chatbot surveys.
Which Players Are Present In The Clinical Trial Site Management Organizations Market Space?
Major companies operating in the clinical trial site management organizations market are IQVIA Holdings Inc., ICON plc, PPD Inc., SGS SA, Syneos Health Inc., Charles River Laboratories International Inc., PAREXEL International Corporation, Medpace Holdings Inc, Premier Research Group Limited, WIRB-Copernicus Group, Worldwide Clinical Trials Inc., ClinChoice Inc., Clinigen Group plc, Precision for Medicine Inc., Synexus Clinical Research, Celerion Inc., Sarah Cannon Research Institute, Clinipace Worldwide Inc., Ergomed plc, Rho Federal Systems Division Inc., Veristat LLC, CTI Clinical Trial & Consulting Services, Clinedge, FOMAT Medical Research INC., Grand Pacific CRO, Xylem CRO, Access Clinical Research LLC, KV Consulting Corporation, Aurum Clinical Research, EthosExcel Inc.
Get The Full Clinical Trial Site Management Organizations Market Report:
Which Region Is The Top Contributor To The Clinical Trial Site Management Organizations Market By Share?
Asia-Pacific was the largest region in the clinical trial site management organizations market in 2025. The regions covered in the clinical trial site management organizations market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Clinical Trial Site Management Organizations Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Clinical Trial Site Management Organizations Market 2026, By The Business Research Company
Clinical Trial Management System Ctms Market Report 2026
Clinical Trial Management Systems Market Report 2026
Clinical Trial Management System Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model