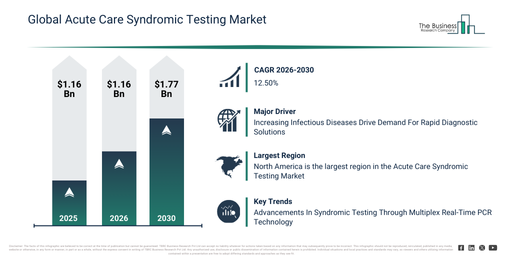
Acute Care Syndromic Testing Global Forecast: Market to Scale Up to $8.29 Billion by 2030 at 12.5% CAGR
Uncover key drivers, emerging technologies, and competitive movements shaping the artificial intelligence (ai) image upscaling chip market from 2026–2035 with trusted insights from The Business Research Company
How much is the Acute Care Syndromic Testing Market valued at in 2026, and what valuation is forecast for 2030?
The market for acute care syndromic testing has witnessed substantial expansion in recent years. This sector is anticipated to increase from $4.59 billion in 2025 to $5.17 billion in 2026, reflecting a compound annual growth rate (CAGR) of 12.8%. Throughout the historical period, this expansion has been propelled by elements like the necessity for emergency infection diagnosis, the enlargement of hospital ICUs, strategies for outbreak readiness, the embrace of molecular diagnostics, and the enhancement of critical care operational processes.
The acute care syndromic testing market size is projected for significant expansion in the coming years. This market is anticipated to reach $8.3 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 12.5%. Key factors driving this growth during the forecast period include AI-enabled syndromic panels, increased emergency testing, antimicrobial stewardship initiatives, advancements in point-of-care molecular systems, and an enhanced focus on pandemic preparedness. Significant trends expected over the same period encompass rapid multiplex pathogen detection, the integration of molecular testing in emergency departments, the development of single-sample multi-disease panels, improvements in clinical decision support speed, and a reduction in diagnostic turnaround time.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=21078&type=smp
What Drivers Are Influencing The Growth Of The Acute Care Syndromic Testing Market?
An increasing occurrence of infectious diseases is anticipated to drive the acute care syndromic testing market in the future. These diseases are defined as ailments resulting from detrimental microorganisms like bacteria, viruses, fungi, or parasites, capable of transmission among individuals, animals, or through the environment. The rise in infectious disease cases stems from factors including population expansion, urbanization, international travel, antimicrobial resistance, and climate change, thereby intensifying the demand for effective prevention and treatment. For infectious diseases, acute care syndromic testing employs multiplex molecular diagnostics to quickly pinpoint pathogens and co-infections, facilitating precise treatment choices. For example, data from February 2024, released by the UK’s Health Security Agency, showed that in week 4 of 2024, the influenza positivity rate climbed to 16.6% from 14.0% in the preceding week. Furthermore, consultations for influenza-like illness (ILI) also saw an increase, reaching 9.8 per 100,000 people, an uplift from 7.5 per 100,000 in the week prior. Similarly, influenza hospital admissions advanced to 7.63 per 100,000, rising from 5.12 per 100,000 the week before. Consequently, the escalating occurrence of infectious diseases is a key factor propelling the acute care syndromic testing market.
How Is The Acute Care Syndromic Testing Market Organized Into Various Segments?
The acute care syndromic testing market covered in this report is segmented –
1) By Disease Type: Respiratory Diseases, Gastrointestinal Diseases, Genitourinary Diseases, Tropical Diseases, Other Disease Types
2) By Sample Type: Blood, Urine, Biofluids, Stool, Swabs, Other Sample Types
3) By Target: Bacteria, Viruses, Fungi, Parasites
4) By End User: Hospitals, Clinical And Diagnostic Laboratories, Research And Academic Institutions, Other End Users
Subsegments:
1) By Respiratory Diseases: Influenza, Respiratory Syncytial Virus (RSV), COVID-19, Tuberculosis, Other Respiratory Diseases
2) By Gastrointestinal Diseases: Clostridium Difficile Infection, Norovirus, Rotavirus, Salmonella, Other Gastrointestinal Diseases
3) By Genitourinary Diseases: Urinary Tract Infections (UTIs), Sexually Transmitted Infections (STIs), Vaginal Infections, Other Genitourinary Diseases
4) By Tropical Diseases: Malaria, Dengue, Chikungunya, Zika Virus, Other Tropical Diseases
5) By Other Disease Types: Meningitis, Sepsis, Other Emerging Infectious Diseases
Which Trends Are Expected To Influence The Acute Care Syndromic Testing Market In The Upcoming Years?
Leading companies within the acute care syndromic testing market are concentrating on technological innovations, such as multiplex real-time polymerase chain reaction (PCR) technology. This focus aims to enhance diagnostic accuracy, facilitate rapid pathogen detection, streamline laboratory workflows, decrease turnaround time, and improve patient outcomes through precise and timely treatment interventions. Multiplex real-time PCR technology is an advanced molecular diagnostic method that simultaneously identifies and quantifies multiple pathogens or genetic targets in a single PCR reaction, thereby increasing efficiency and accuracy in disease diagnosis. For example, in April 2023, QIAGEN N.V., a Netherlands-based biotechnology research company, introduced the QIAstat-Dx syndromic testing solution in Japan. This solution features a SARS-CoV-2 respiratory panel designed to detect over 20 pathogens from a single sample. Utilizing multiplex real-time PCR technology, it delivers quick results with minimal labor requirements compared to individual PCR assays. This system is available in over 100 countries, consequently enhancing syndromic testing accessibility across the globe.
Who Are The Primary Competitors In The Global Acute Care Syndromic Testing Market?
Major companies operating in the acute care syndromic testing market are Siemens Healthineers AG, F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific Inc., Danaher Corporation, Becton Dickinson and Company, Seegene Inc., bioMérieux SA, Sysmex Corporation, Bio-Rad Laboratories Inc., QIAGEN N.V., Werfen, DiaSorin S.p.A., Quidel Corporation, Luminex Corporation, Biocartis NV, Microbiologics Inc., QuantuMDx Group Ltd., Curetis GmbH by OpGen Inc., Genetic Signatures Ltd., Applied BioCode Inc.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/acute-care-syndromic-testing-global-market-report
Which Global Regions Are Shaping The Competitive Landscape Of The Acute Care Syndromic Testing Market?
North America was the largest region in the acute care syndromic testing market in 2025. The regions covered in the acute care syndromic testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Acute Care Syndromic Testing Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=21078&type=smp
Browse Through More Reports Similar to the Global Acute Care Syndromic Testing Market 2026, By The Business Research Company
Tele Intensive Care Global Market Report 2026
https://www.thebusinessresearchcompany.com/report/tele-intensive-care-global-market-report
Severe Acute Respiratory Syndrome Treatment Global Market Report 2026
Tele Intensive Care Unit Global Market Report 2026
https://www.thebusinessresearchcompany.com/report/tele-intensive-care-unit-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company