Cablivi Market Forecast 2026–2035: How to Leverage Market Growth
Uncover key drivers, emerging technologies, and competitive movements shaping the cablivi market from 2026–2035 with trusted insights from The Business Research Company
What is the expected shift in the Cablivi Market’s size between 2026 and 2030?
The expansion observed during the historic timeframe can be ascribed to multiple factors. These include the substantial mortality rate among individuals with untreated attp, the inherent constraints of relying solely on plasma exchange, the subsequent approval of specific vwf inhibitors, a heightened understanding of the disease, and the broader integration of specialist hematology practices.
Anticipated expansion during the forecast period is fueled by factors such as earlier detection of attp, the proliferation of specialized hematology centers, enhanced access to targeted biologic treatments, robust reimbursement structures, and continuous innovation in rare blood disorders.
Key developments expected within the forecast timeframe include the expanding utilization of targeted therapies for attp, the increasing embrace of nanobody-based pharmaceutical products, a strong emphasis on achieving swift disease control, the broadening array of treatments for rare hematologic conditions, and advancements in acute care management.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=20009&type=smp
Which Drivers Are Shaping Strategic Decisions In The Cablivi Market?
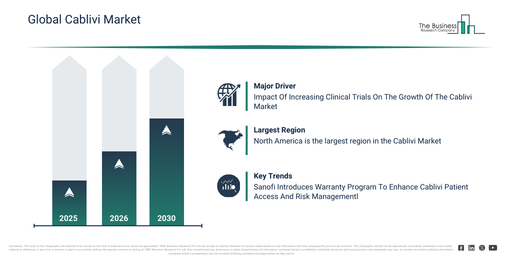
The cablivi market is anticipated to expand due to the increasing number of clinical trials. These trials are research studies involving human participants to assess the safety, effectiveness, and efficacy of medical treatments, interventions, drugs, or devices. The rise in clinical trials stems from advancements in medical research and technology, the growing incidence of chronic and complex diseases, and an aging global population demanding new treatments. Cablivi benefits from clinical trials by providing robust evidence of its efficacy and safety in treating Thrombotic Thrombocytopenic Purpura (TTP), which has contributed to its regulatory approval and widespread use in clinical practice. For instance, in February 2024, data from ClinicalTrials.gov, a US-based government web resource, showed that clinical trials publishing findings rose from 4,024 in 2022 to 5,063 in 2023. Consequently, the increasing volume of clinical trials is driving the growth of the cablivi market.
What Segment Categories Are Covered In The Cablivi Market?
The cablivi market covered in this report is segmented –
1) By Clinical Indication: Acquired Thrombotic Thrombocytopenic Purpura (aTTP), Congenital Thrombotic Thrombocytopenic Purpura (cTTP)
2) By Distribution Channel: Direct Sales, Pharmaceutical Wholesalers, Online Pharmacies, Retail Pharmacies
3) By End Use: Hospitals, Clinics, Ambulatory Care, Home Care
Which Trends Are Influencing The Development Of The Cablivi Market?
Leading companies operating within the Cablivi market are focusing on developing advanced solutions, such as warranty programs, to enhance patient access and reduce financial risk. These warranty programs are structured initiatives designed to refund the cost of treatment for patients who do not achieve an initial clinical response or who experience a worsening of their disease. For instance, in January 2023, Sanofi SA, a France-based pharmaceutical and healthcare company, introduced a warranty program for Cablivi, its medication for acquired thrombotic thrombocytopenic purpura in adults. The program offers coverage for up to six inpatient doses if patients do not reach an initial clinical response, and up to 12 doses for those whose condition exacerbates during treatment. Cablivi is administered concurrently with plasma exchange and immunosuppressive therapy to prevent dangerous blood clots. It represents Sanofi’s first nanobody-based medicine and works by targeting von Willebrand factor to inhibit its interaction with platelets. The launch of this program underscores key features including reimbursement tied to clinical response, coverage for inpatient doses, support for hospital-administered therapy, and strategies for mitigating the risks associated with high-cost treatment.
Which Major Firms Are Strengthening Their Position In The Cablivi Market?
Major companies operating in the cablivi market are Sanofi S.A.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/cablivi-global-market-report
Which Region Is Anticipated To See The Fastest Growth In The Cablivi Market?
North America was the largest region in the cablivi market in 2025. The regions covered in the cablivi market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Cablivi Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=20009&type=smp
Browse Through More Reports Similar to the Global Cablivi Market 2026, By The Business Research Company
Undersea Cabling Market Report 2026
https://www.thebusinessresearchcompany.com/report/undersea-cabling-global-market-report
Trade Finance Market Report 2026
https://www.thebusinessresearchcompany.com/report/trade-finance-global-market-report
Commercial Coffee Urn Market Report 2026
https://www.thebusinessresearchcompany.com/report/commercial-coffee-urn-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model